Regulatory Publishing
Automation
for Pharma Biotech
Document Publishing is important, and companies optimize this by best practices, outsourcing and some tools. Doesn’t matter what tools or vendors you use currently, probably most of the steps or activities are still largely manual. This may be leading to reworks, longer cycle times, people burn out, additional costs and frustrations. Whether your source files are in MS WORD or PDF, get submission ready files in minutes.
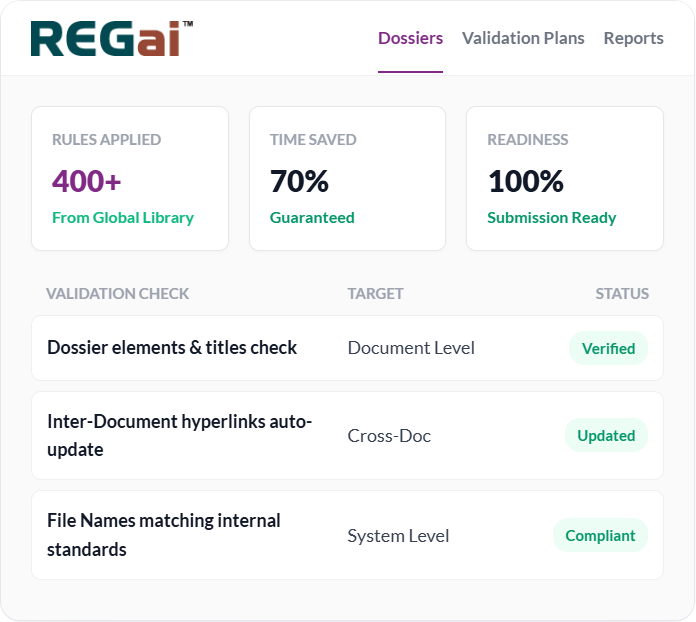
0%+
Time & Cost Savings
0+
Rules Library
0%
Submission Ready
3
Flexible Deployment Options
The Challenge: Limits of Traditional
Publishing
Even with outsourcing and existing tools, document publishing can be a bottleneck. Most steps
or activities remain largely manual, compromising speed and quality.
Longer Cycle Times
Manual alignment of elements, formatting, and standardizations cause unnecessary delays in critical submission timelines.
Reworks & Errors
Human interventions naturally lead to mistakes, forcing repetitive reworks and adding stress to regulatory teams.
Team Burnout
Constantly dealing with tedious validation and manual hyperlink updates creates significant frustration and team fatigue.
Additional Costs
The accumulation of delays, manual labor, and rework pipelines drive up overall publishing costs substantially.
Our Solution:
Efficient Automated Publishing
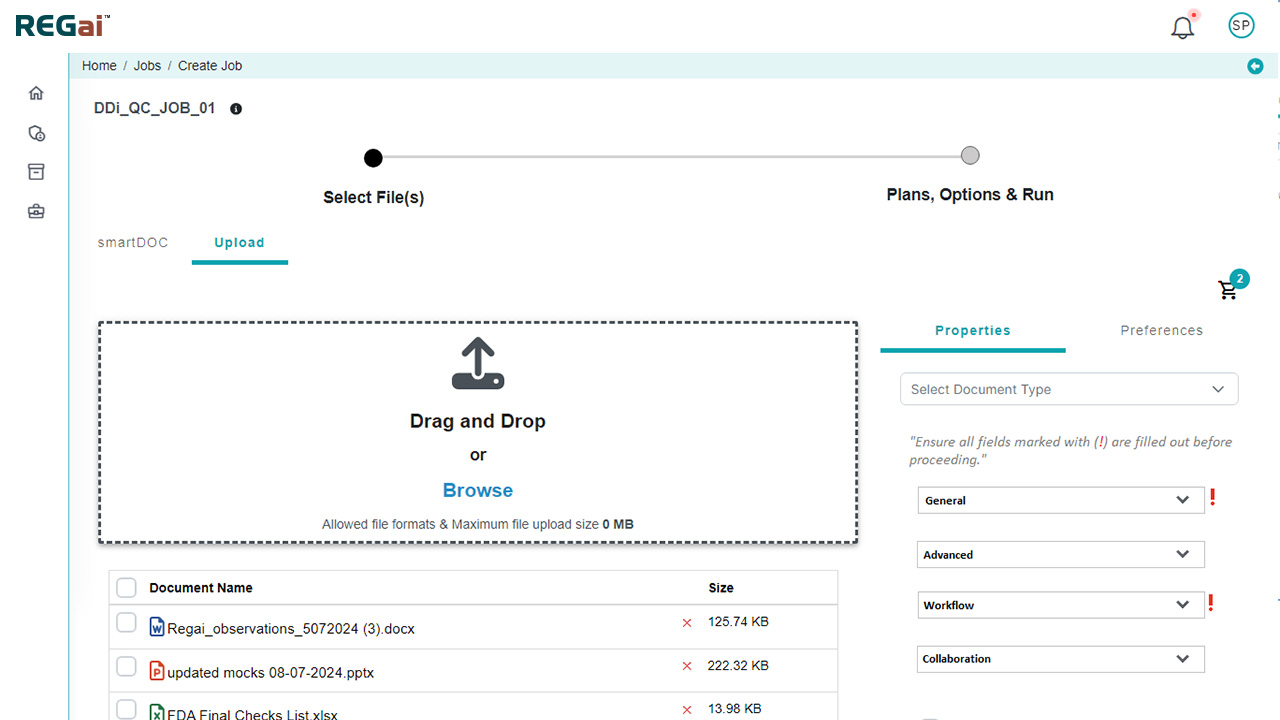
Our solution automates document publishing without disturbing your current eCTD infrastructure. It aligns to your current systems and processes by providing different options (local, cloud, plugins, integration).
With a deep 400 rules library, you pick and create your custom plans with the flexibility of applying what and when to your documents.
Word Plugins
Deploy seamless publishing checks directly within your MS Word environment.
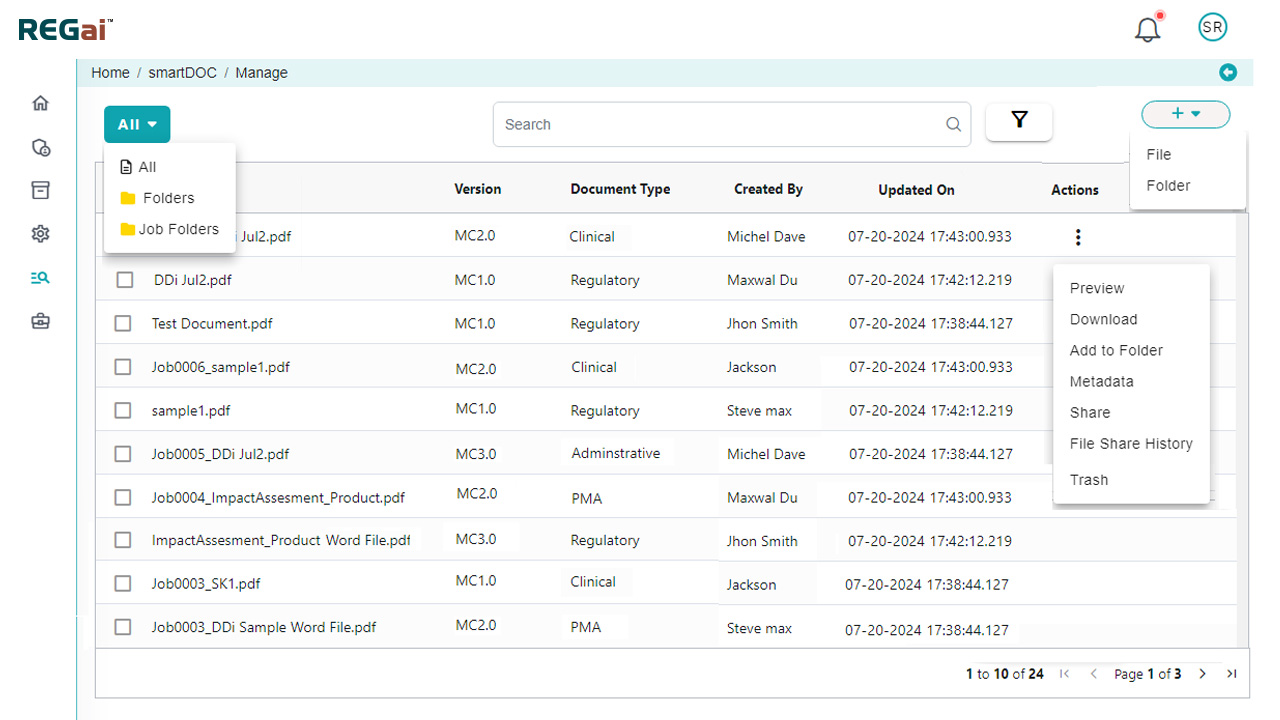
Desktop / EDMS Uploads
Use our standalone tool to effortlessly upload and process files from your desktop or EDMS.
Batch Processing
Connect to existing Document stores and perform automated publishing on your schedule (hourly, daily).
Pre-Built Interfaces & Post-Publishing
We seamlessly integrate with your current eCTD systems, RIM, EDMS, ERP, SharePoint, Documentum, BOX, and other 3rd party Sources.
Once connected, you can automate validation with customized Dossier checks covering dossier elements, titles, and data/content level based on your specific checklists.
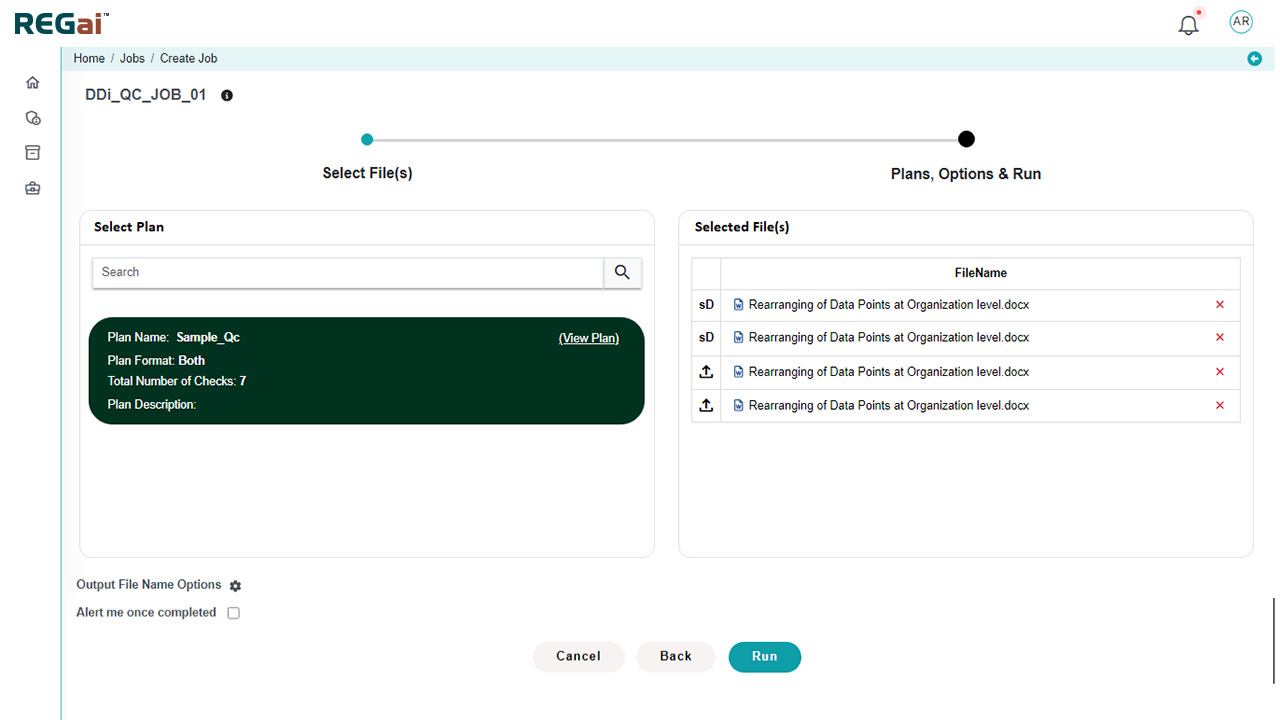
Easily create custom validation plans at the dossier level and selectively test validation paths for standard and custom dossiers.
Inter-Document Hyperlinks
Auto-Update File Names
Custom Validation Plans
Leaf Titles Update
Key Capabilities
A comprehensive library of rules and integrations to streamline your publishing.
Source Flexibility
Whether your source files are in MS WORD or PDF, process them efficiently to get submission ready files in minutes.
400+ Rules Library
Leverage a deep repository of 400 rules to create customized plans and apply automated checks when needed.
Seamless Interfaces
Pre-built interfaces to connect directly with RIM, EDMS, SharePoint, BOX, Documentum, and eCTD systems.
Post-Publishing Validation
Automate validation covering dossier elements, titles, and data content based strictly on your checklists.
Hyperlink Automation
Automatically check, generate, and update critical Inter-Document hyperlinks across your entire submission.
Naming Standardizations
Ensure compliance through auto-updating of File Names, Elements, and Leaf titles matching your internal standards.
Guaranteed Results & Benefits
Transform your document lifecycle and optimize your regulatory submissions.
70%+ Time & Cost Savings
Drastically cut down your current document publishing costs and cycle times. Guaranteed.
Eradicate Manual Efforts
Remove the tedious, repetitive tasks that lead to team burnout, freeing resources for strategic work.
Zero Reworks
Rely on 400+ automated rules and custom validation plans to get the dossier right the first time.
Zero Infrastructure Disturbance
Our solution aligns perfectly with your current systems—offering local, cloud, or plugin deployments without disruption.
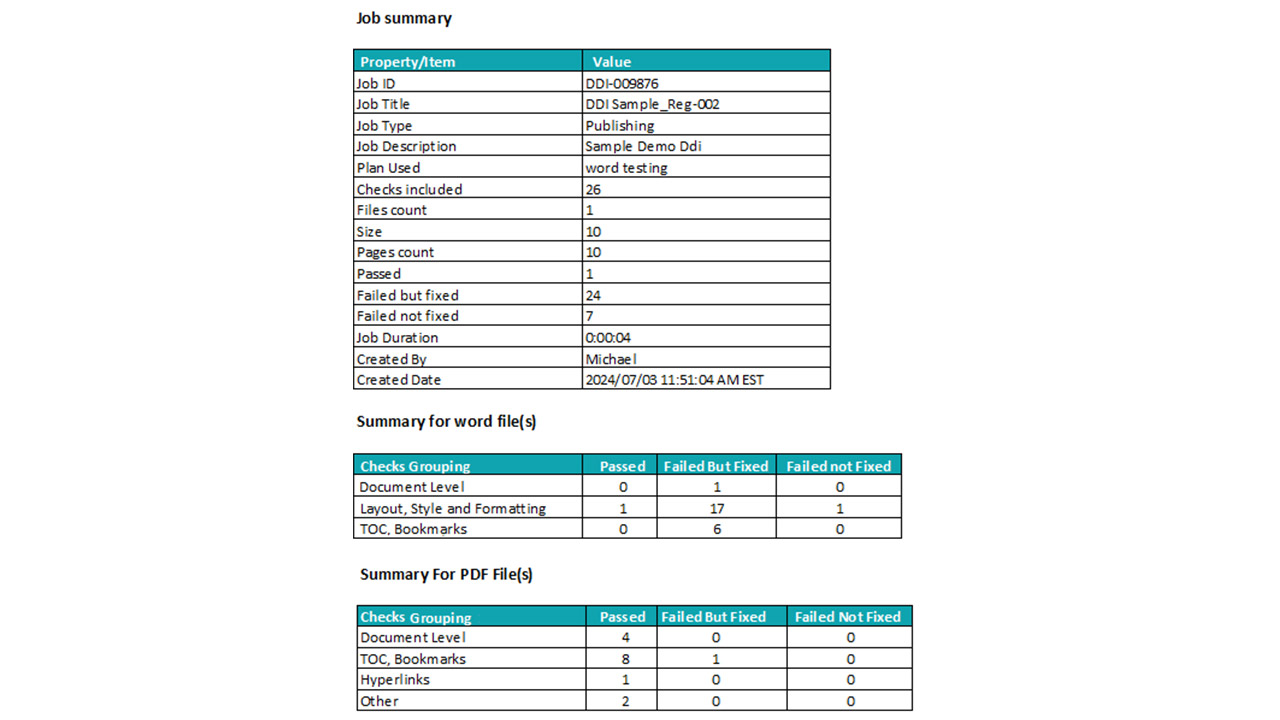
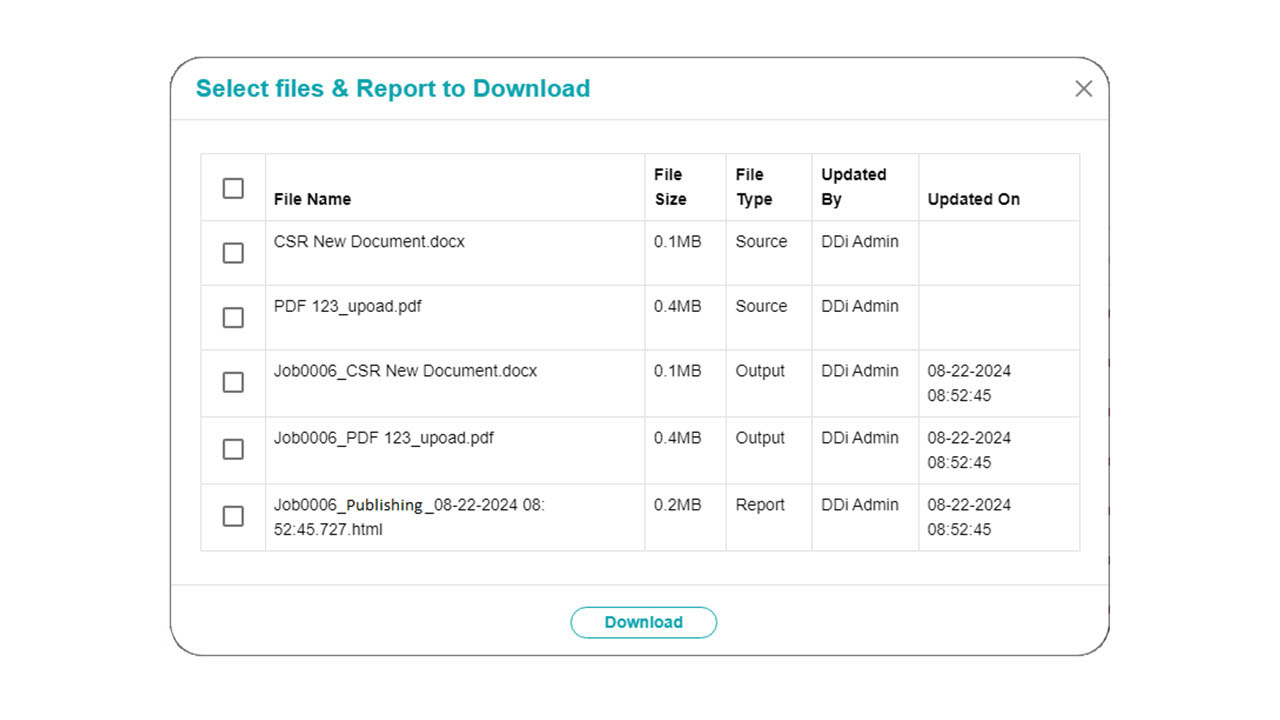
Platform Interface
Experience the intuitive design of DDi's Publishing Automation Solution.





Ready to Automate Your Publishing?
Reduce manual efforts, reworks, and cycle times. Get your submission-ready
files in minutes with guaranteed cost savings.
Related Tools & Solutions

Submission Management

Regulatory Information Managemen...

Regulatory Document Management
Explore Insights

QC Bottleneck in Publishing: How Automation is Solving

Technical Documents: Accelerate with Agentic Automation for MedTech

Hyper-Efficient Regulatory Publishing: Agents & Automation
Let's talk about how DDi can help you