RTSM / Clinical Supplies IRT
Why Supplies Management matters?
In an era of improved regulations to guarantee patient safety, clinical trials have become increasingly complex. Study sponsors now aim to meet multiple objectives and as the level of complexity grows, clinical supplies strategy should be supported by technology to facilitate the process at the site and depots. At the same time systems should offer more visibility across the supply chain, leading to better planning while maintaining regulatory compliance. With Direct-to-patient as a trend that will stay, systems need to have these built-in and to become standard option going forward.
mIRT optimizes clinical trial supply management practices to help position your trials towards success.
Advanced Technology
Cloud-based, easily configurable, and advanced integration features (can be integrated with data from wearable) enabling real-time access to study data and admin tools through the mobile interface and monitoring your study’s advancement to make important decisions.
Tech-Driven Inventory Management
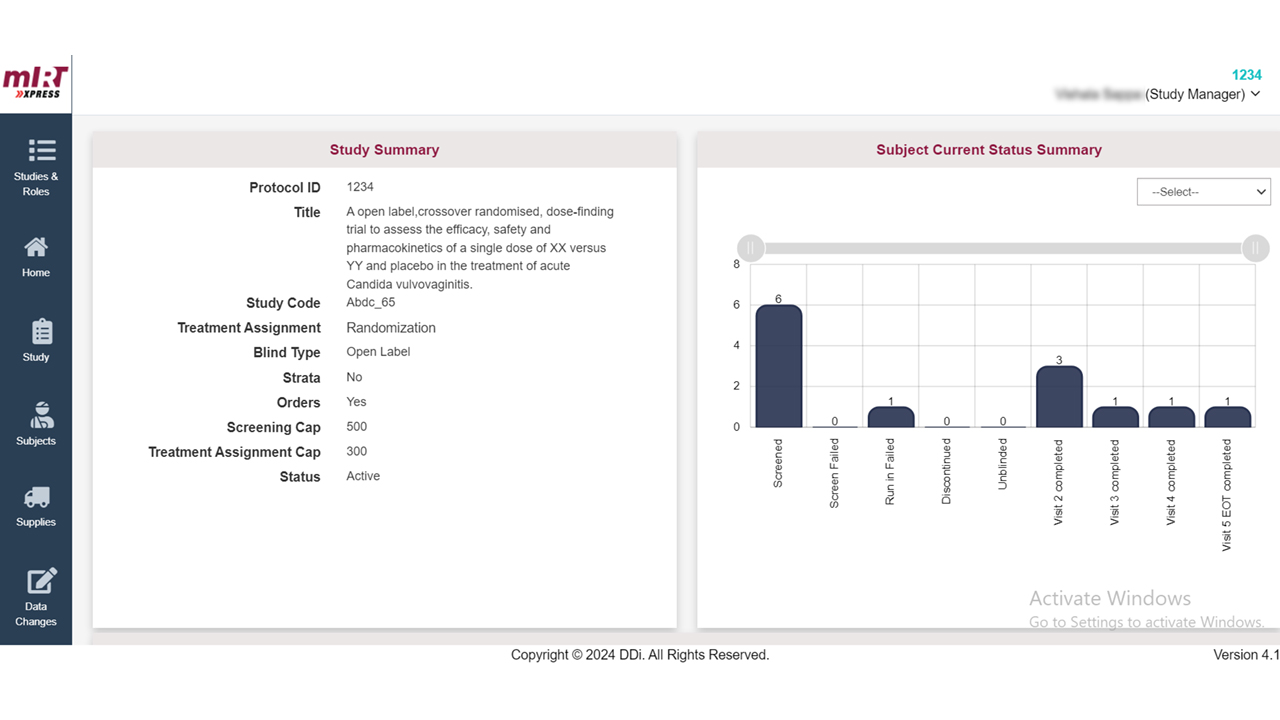
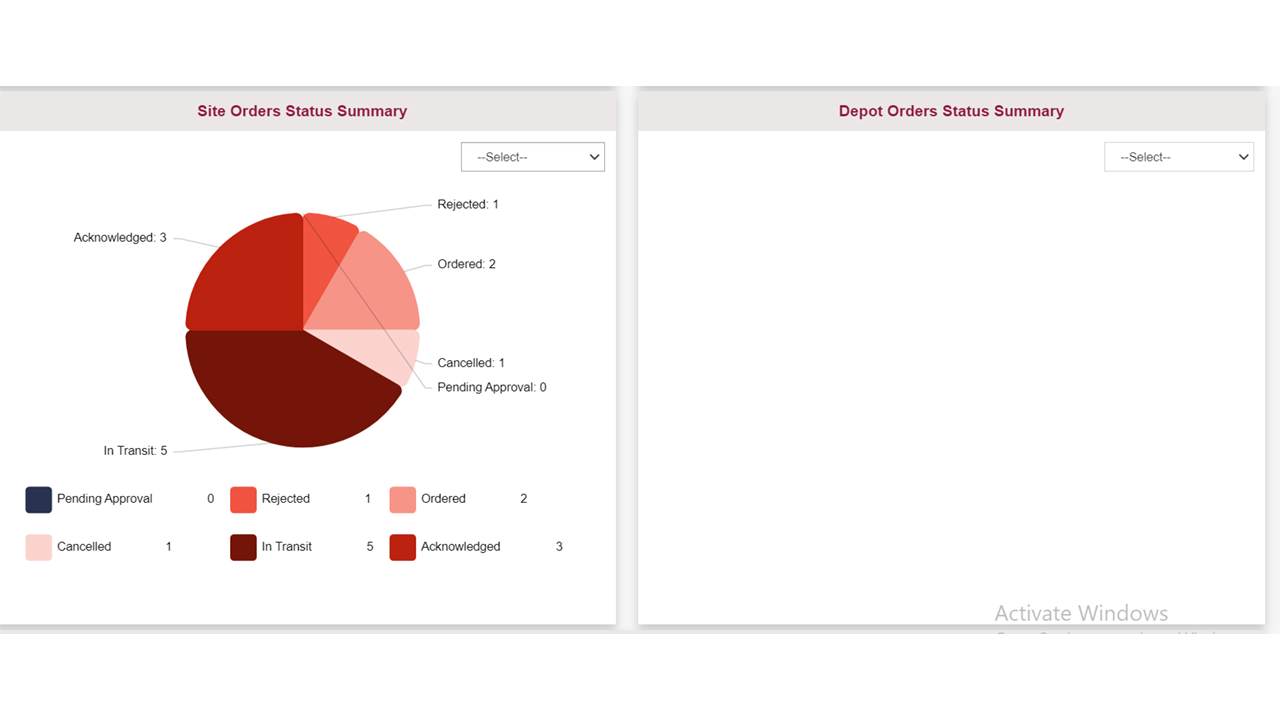
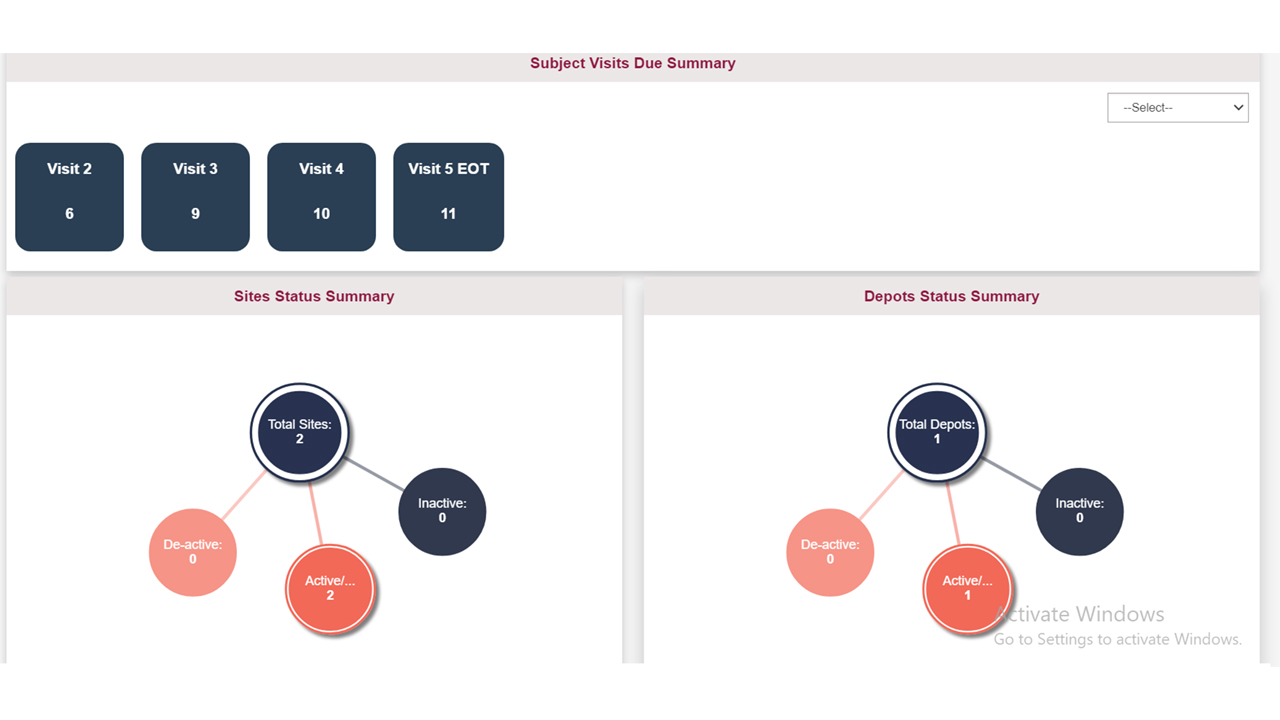
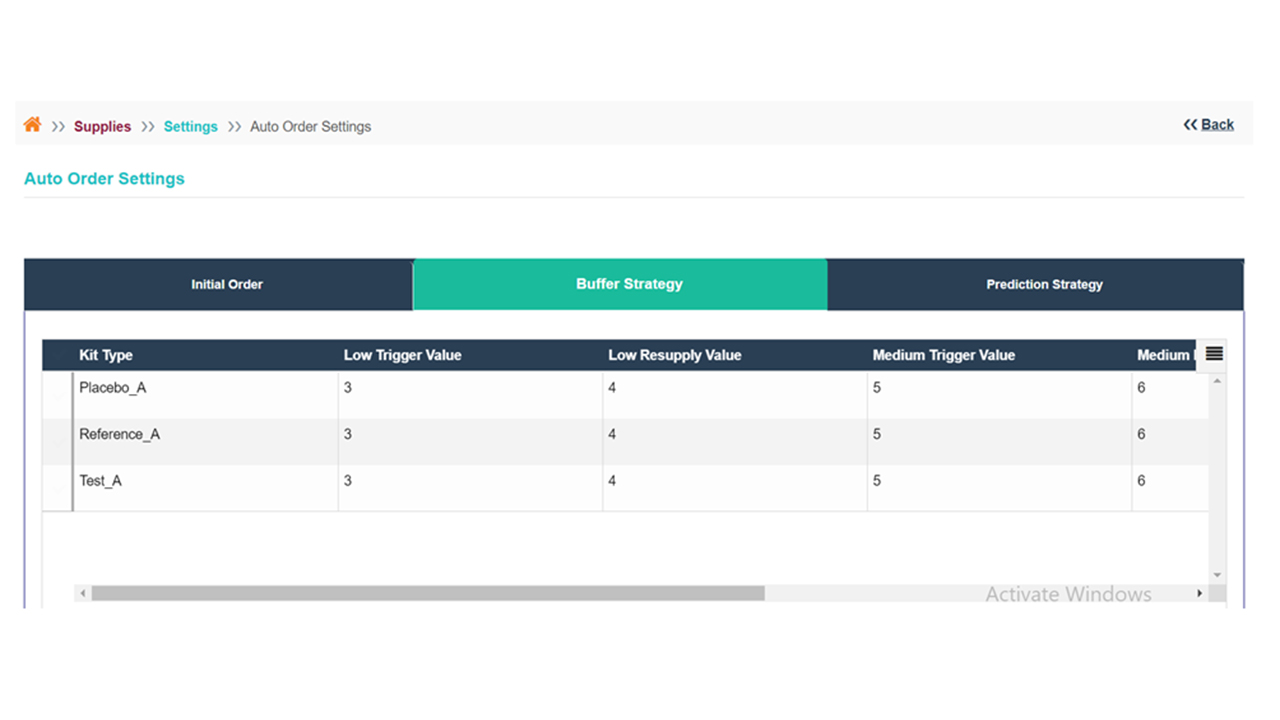
Our solution offers a real-time view of supplies during a clinical trial, enabling users to efficiently plan and manage supplies in a single centralized location. Automated and customizable Tech-driven supply/re-supply schemes to track and optimize the trial’s timeline and stay on top of resupply.
Trial Supply Intelligence
Utilizing our cross-study custom reporting modules with analytics gives you visibility on inventory across all studies. Search, filter, and export supply figures or upcoming expiring supplies at sites in one central location.
Adaptive & Complex Trials
mIRT supports adaptive and complex protocol designs with configurability, drug pooling strategies, and program-based supply management. mIRT makes it easy to manage cohorts, add/delete new treatment arms or kit types, allocate or add drug supplies across multiple trials, and more.
No matter the complexity of your protocol or clinical supply strategies, mIRT defines solutions that:
-
Reduce risks in your trial
-
Optimize your supply chains
-
Ensure continuous drug supply
-
Make mid-trial changes easier and faster
-
Increase start-up speed
-
Boost productivity and improve data quality
-
Reduce overall cost of trial drug management
We’re Here To Help
Get in touch with us
Let's talk about how DDi can help you

