
Paperless Compliance.
Global Reach.
The proven e-labeling solution for Medical Devices & IVDs. Minimize risk, save costs, and ensure compliance with ISO
13485, EU MDR, FDA, and global regulations.
0+
Years Experience
27001
ISO Certified Security
13485
ISO Certified Quality
0%
Audit Ready
Trusted by Leading Medical Device Companies











Why Choose Visu eIFU?
An end-to-end, compliant, and user-centric eIFU solution designed to simplify global regulatory requirements.
Global Compliance
EU MDR, FDA 21 CFR Part 11, Canada, Brazil, and Australia compliance included. We track country-specific regulatory changes for you.
Highly Configurable
An intuitive, off-the-shelf solution with a highly configurable interface and smart search functionality tailored to your device portfolio.
Robust Security
Data hosted in ISO 27001 certified data centers (USA/Germany). Full GDPR compliance with audit-ready documents and validation.
Print Services
Integrated ISO 20000-compliant service desk managing print requests with a guaranteed 7-day turnaround per EU regulations.
Analytics & Audits
Comprehensive backend analytics, role-based access control, and full audit trails for traceability and self-service reporting.
User Centric
Control what healthcare professionals see versus patients. Manage content by country and language with subscription notifications.
Key Capabilities
Why medical device manufacturers choose Visu eIFU for their digital transformation.
Out-of-box or Custom eIFU Portal
Visu is designed for speed and simplicity – get your eIFUs online compliantly without hassle.
- Intuitive, off-the-shelf solution
- Easy to implement
- Highly configurable interface
- Smart search functionality
- Personal training on use of Visu eIFU
- Expert regulatory support
- Trusted by Notified Bodies
Regulatory Compliance
Compliance is core of Labeling. Constant checking of different country changes and informing manufacturers on changes.
- EU MDR, FDA, Canada, Brazil, Australia compliance included
- FDA 21 CFR Part 11 compliant
- Analytics and reporting features
- GDPR compliance
- All audit-ready documents provided
- Data centre certified in ISO 27001
Admin Access for You & Controlled Document Access to End Users
Manage your eIFUs with ease using Visu’s secure, compliant, and customizable admin platform.
- Role-based access control for manufacturers
- Workflow based and edms inbuilt
- All Reports along with Audit trail included in backend for self-service
- For end users, control or manage what healthcare professions should see and what patients should get
- Mange by country and language
- Subscription & notification
- Enterprise edition: PLM/EDMS integration, workflows, branding
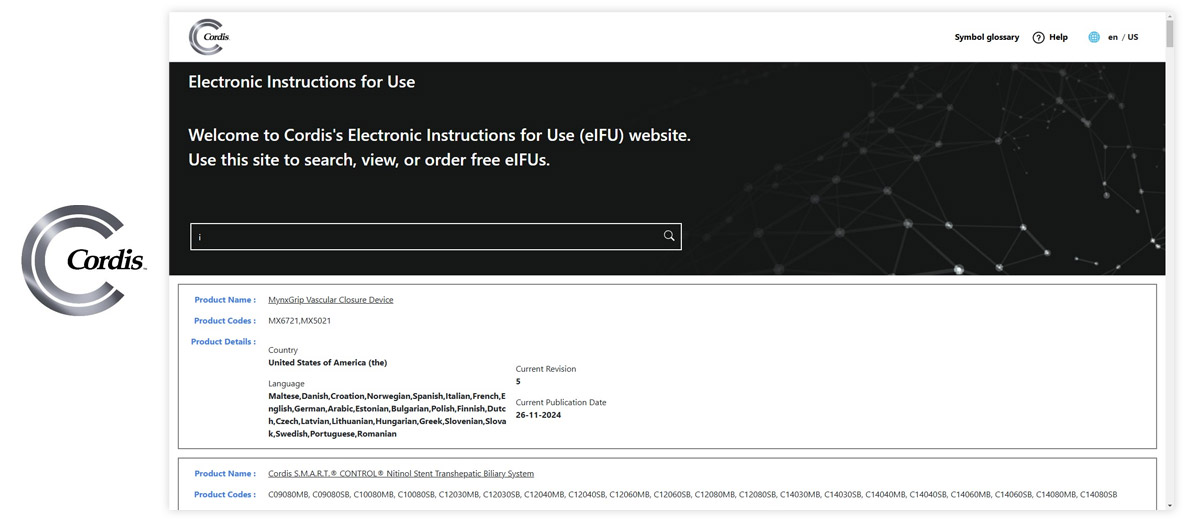
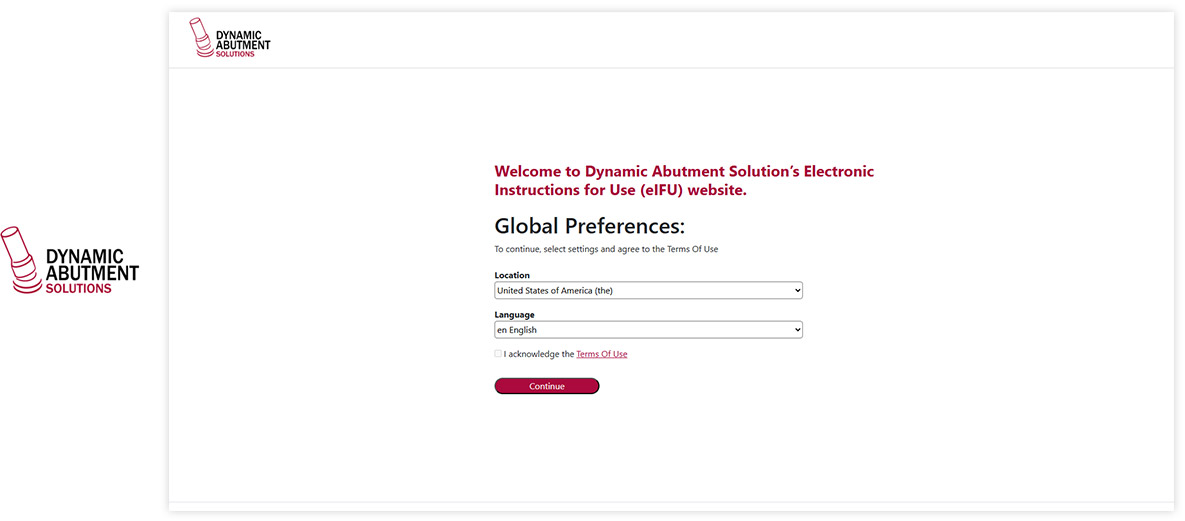
Made with Visu eIFU Platform
See how leading MedTech brands present their e-labeling portals. (Click images to enlarge)
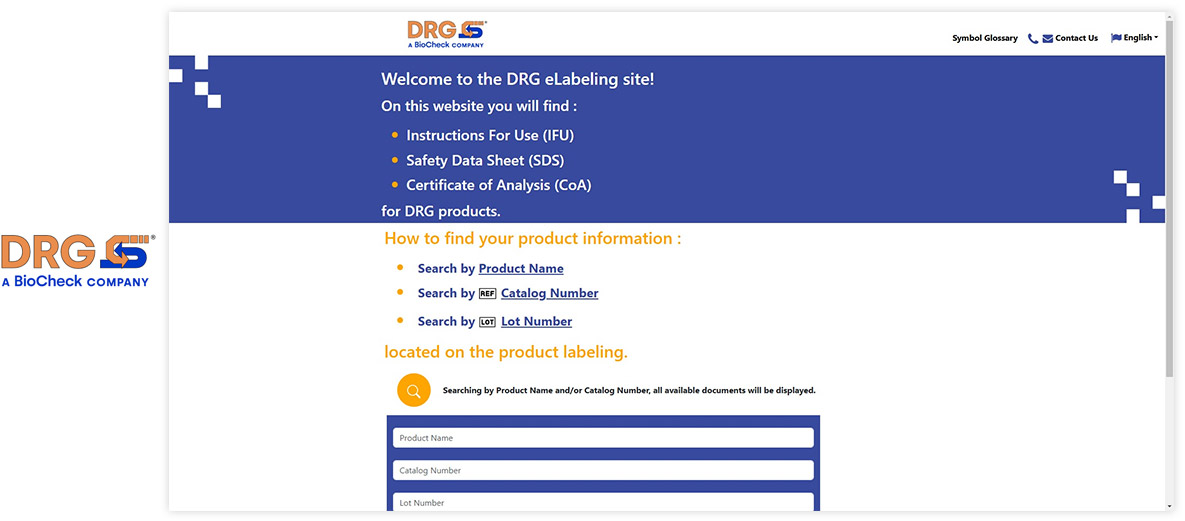
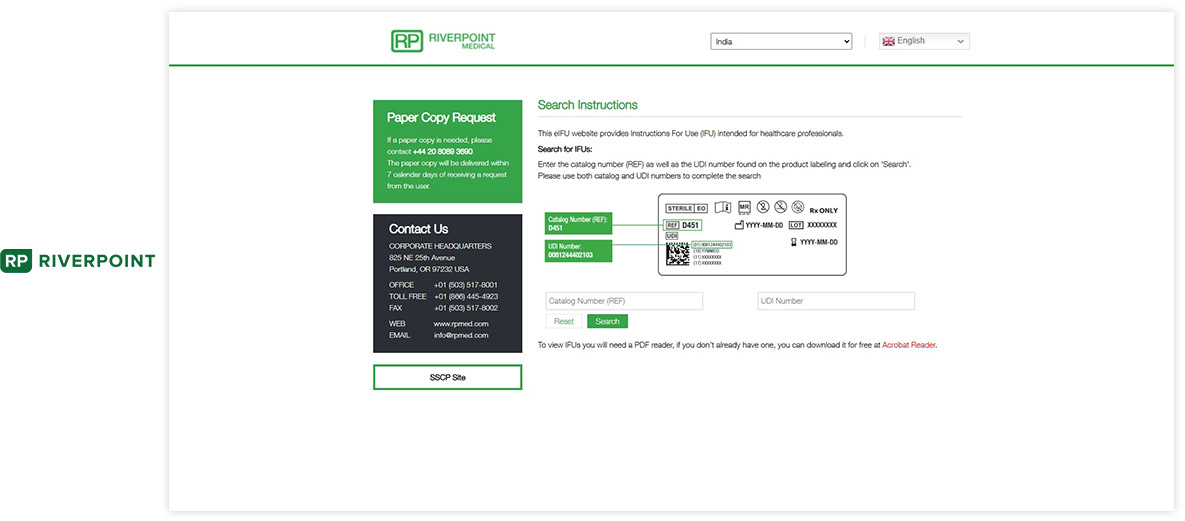
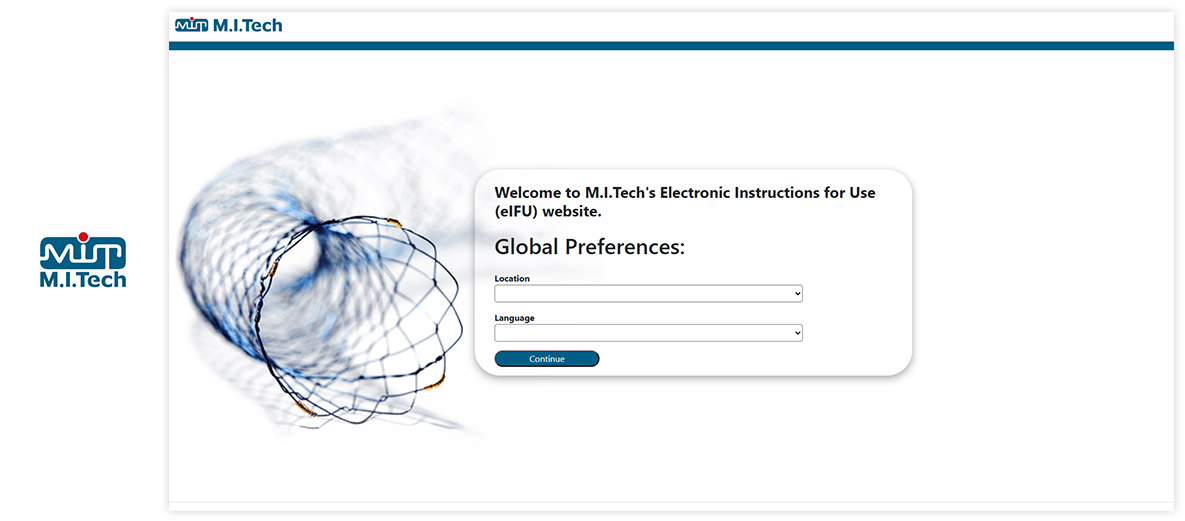





Ready to Digitize Your IFUs?
Experience seamless e-labeling with Visu eIFU - ensuring compliance, global access, and user-friendly navigation.
Frequently Asked Questions (FAQs)
Related Tools & Solutions

UDI Solutions

Regulatory Information Managemen...

EUDAMED Software and Consulting...
Explore eIFU Insights

Navigating the Global eIFU Patchwork: EU, USA, and the ASEAN P...

AI-Driven Workflows for Medical Devices | R&D to Compliance

Electronic Instructions for Use (eIFU): The Complete 2026 Guid...
eIFU Regulations & Guidelines
Regulations and Guidelines
eIFU guidance for IVDs
eIFU regulation for MDs (under MDR)
eIFU regulation for MDs (under MDD)
EU regulation for IVDs
EU regulation for MDs
GDPR
MEDDEV 2.14/3 rev.1 Supply of Instructions For Use (IFU) and other information for In-vitro Diagnostic (IVD) Medical Devices.
Read more:
Commission Implementing Regulation (EU) 2021/2226 of 14 December 2021 laying down rules for the application of Regulation (EU) 2017/745 of the European Parliament and of the Council as regards electronic instructions for use of medical devices.
Read more:
Commission Regulation (EU) No 207/2012 of 9 March 2013 on electronic instructions for use of medical devices.
Read more:
Regulation (EU) 2017/746 of 5 April 2017 on In Vitro Diagnostic Medical Devices and Repealing Directive 98/79/EC and Commission Decision 2010/227/EU.
Read more:
Regulation (EU) 2017/745 of 5 April 2017 on Medical Devices, amending Directive 2001/83/EC, Regulation (EC) No 178/2002 and Regulation (EC) No 1223/2009 and repealing Council Directives 90/385/EEC and 93/42/EEC.
Read more:
Regulation (EU) 2016/679 of 25 May 2018 on Regulation on the protection of natural persons regarding the processing of personal data and on the free movement of such data, and repealing Directive 95/46/EC (Data Protection Directive).
Read more:
US Regulations (FDA)
FDA regulation for MDs & IVDs
FDA regulation for MDs & IVDs on labeling and advertisements
FDA regulation on use of electronic records & signatures
FDA regulation to authorize eIFU
Federal Food, Drug, and Cosmetic Act (FD&C Act) – Chapter V: Drugs and Devices – Part A Drugs and Devices and Part D Dissemination of Treatment Information.
Read more:
U.S. Code, Title 21, Chapter 9, subchapter V, Part A, §352 Misbranded drugs and devices.
Read more:
U.S. Code, Title 21, Chapter 11, Electronic records; electronic signatures.
Read more:
Section 206 of the Medical Device User Fee and Modernization Act (MDUFMA) (New section 502(f) of the Federal Food, Drug, and Cosmetic Act) Electronic Labeling for Prescription Devices Intended for Use in Health Care Facilities.
Read more:
US Guidance Documents (FDA)
FDA regulation for MDs & IVDs
Guidance for Industry and FDA Staff: Acceptable Media for Electronic Product User Manuals
Guidance for Industry; March 2006
Read more:
March 2010.
Read more:
Notice 10-123767-875: Guidance for the labelling of Medical Devices, not including in vitro diagnostic devices.
Read moreNormative Instruction – IN No. 4, of June 15th 2012.
Read moreSFDA/MDS Guidance on Requirements for Electronic Instructions for Use (e-IFU) of Medical Devices (MDS – G41, of September 29, 2019)
Electronic Instructions for Use – eIFU: For professional users of medical devices (including IVDs) – v1.0 August 2018 (Therapeutic Goods Administration).
Read moreSDLC standard used for the development of the IFUcare website
IEC 62304:2006: Medical device software – Software life cycle processes
ISO 13485
Medical Devices - Quality Management Systems - Requirements for Regulatory Purposes.
ISO 14971
Medical Devices – Application of Risk Management to Medical Devices
Let's talk about how DDi can help you