UDI – Comply with ALL countries with ONE Solution
Unique Device Identification (UDI) is a key global harmonization process for more reliable data that aids overall medical device traceability throughout the device supply chain. Health Authorities rolled out medical device UDI requirements, such as GUDID by US FDA, EUDAMED by European Union under new MDR/IVDR, NMPA UDI by China, and many more. Global device manufacturers must often submit UDI data to a several of country-specific regulatory authorities, each with their unique data exchange standards and requirements.
Visu UDI takes a platform approach to help you overcome several drawbacks and short comings of solutions like GDSN, PLM/ERP based modules, and custom bespoke solutions. As countries requiring UDI increasing, you need a solution that’s flexible and adapts to continuous changes (both your internal data and external country requirements) without burdening internal resources and ensure keeping UDI compliance cost in control.
Our UDI solution enables device manufacturers to implement a single data syndication system for global coverage. Leveraging one solution to aggregate, validate and submit product data to regulatory agencies at the market-specific level around the globe, all done in cloud (or can implement on your servers).
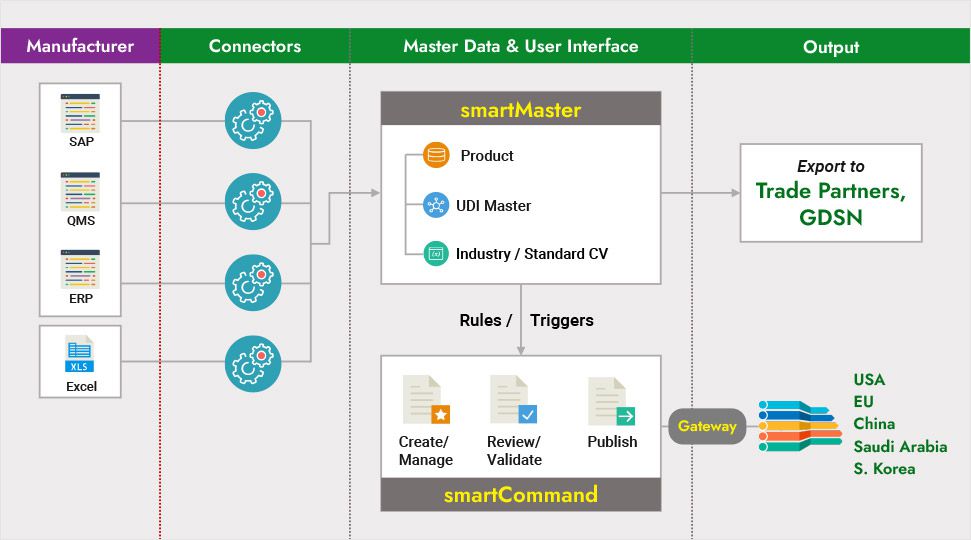
Snapshot of our Solution with 2 major pieces – smartMaster, smartCommand
Functionality

Workflow and User Interaction
Leverage drag-and-drop workflow creation to meet any processes you have currently to ensure make the overall process as “Interactive” as possible or as “batch / backend run” as possible.
Customize / Configure triggers to link different data changes to your internal processes and actions along with alerts when user intervention is required, either for error issues or business rules exceptions.

Data Pooling & Mapping (smartMaster)
With built-in 3rd party connectors, data interface can be developed easily (or can do custom interfaces if you have legacy or internal tools). In addition, system supports other formats like csv, xml inputs as well. ‘UI based Mapping” functionality helps data analysts map, extract and collate master data to any source data recipient, be it a system or a process.
All major file & message formats are supported ‘out-of-the-box’. Data collation can be automated using scheduled jobs, or real-time web services APIs.

Validation
Deliver trusted, high-quality product data by leveraging our standard data quality and data validation tools.
Ensure that product data is accurate and compliant with any pre-defined set of validation rules from regulatory agencies, as well as any internal, user-configured data attributes and validation rules.

Electronic Submissions / Gateway
Proven electronic submission capabilities for regulatory data submissions that ensure data accuracy and a scalable process for data compliance in all markets.
Ease of enabling new countries at a fraction of the cost of alternatives such as data submission services or standalone country-level compliance solutions.

UDI Interfaces
Pre-configured connectors to GDSN-compliant data pools that enable global data synchronization with recipient-specific product attributes.
Out-of-the-box connectors to UDI databases across the globe including USA, EU, China, S Korea, Saudi Arabia and others are in development currently.

System Integration
Easily integrate to RIM, PLM, ERP and other systems to source the relevant master data using configurable message/file formats.
Supports AS2 gateways and AS4 Access Points for message delivery using all required XML formats including the GS1 XML for GDSN, HL7 SPL for GUDID, and SOAP XML for EUDAMED.
Global Specific UDI Requirements by Countries
- Australia (TGA)
- Brazil (ANVISA)
- Canada (Health Canada)
- China (NMPA)
- European Union (EC)
- Japan (PMDA)
- Saudi Arabia (SFDA)
- Singapore
- South Korea (MFDS)
- Taiwan
- UK (MHRA)
- United States (FDA)
-
- 2022 Jun – AUSUDID sandpit (beta) — general use, testing
- 2023 Jan – Regulations and Guidance — in effect
- 2023 Jan – Voluntary Compliance (high risk Class II, III, AIMD)
- 2024 – Mandatory Compliance (implants)-UDI labeling, UDI data (Implementation timeframes and compliance dates will be published as soon as they are confirmed)
- Approach: similar to EU regulation; UDI/Device registration and UDI labels implemented by MD and IVD classes
- UDI Data: ‘EU-like’ attributes reported to AusUDID
- Data Sub: Bulk upload via Microsoft Excel files, Machine-to-Machine (M2M) upload via HL7 SPL files and Data transmission via the National Product Catalogue (NPC)
- UDI Label: HRI & AIDC; Direct Mark
- STD: GS1/HIBCC/ICCBBA, EMDN/GMDN?
- Info: Medical Device & IVD
-
- 2020 Jun – UDI Barcode inserts required in a few implant product packages; UDI data reported to RNI by healthcare provider
- 2021 Feb – UDI System Publication
- 10 July 2025-Class IV (highest-risk) devices
- 10 January 2026-Class III (high-risk) devices
- 10 January 2027-Class II (Moderate-risk) devices
- 10 January 2028-Class I (Low-risk) devices
- Approach: Pilot started with package UDI barcodes on Coronary Artery Stents and Hip & Knee Arthroplasty Implants
- UDI Data: Few attributes reported by healthcare provider to National Implant Registry (RNI)
- UDI Label: DI, Expiry Date, Lot/SN in AIDC (1D or 2D barcode) on (3) Mfr-supplied “Traceability Labels” with product pkg
- STD: GS1/HIBCC,ICCBBA
- Info: ANVISA (National Health Surveillance Agency)
-
- 2022 – Proposal Health Canada UDI
- Approach: Ongoing UDI preparation, expected to follow IMDRF UDI recommendations
-
- 2019 2H – UDI Pilot, Rules, UDID database tests
- 2019 Sept 17 – Batch 1 Notice timing and list
- 2020 Sept 30 – Batch 1 Delay and 69 (64+5) categories
- 2021 Jan 01 – Batch 1 UDI Data & Label Required
- 2022 Jun 01 – Batch 2 (Remaining Class III)
- 2024 Jun 01 Class II
- 2026 – Class I and Remaining Devices
- Website upload or Batch import
- GS1, ZIIOT and AHM.
-
- 2020 Dec 01 — Actor Voluntary Economic Operator Registration
- Currently EUDAMED is voluntary, and its mandatory use date has been postponed multiple times.
- Placing UDI-carriers on the labels of Medical devices
- 26 May 2021 – Implantable and class III devices
- 26 May 2023 — Class IIa and class IIb devices
- 26 May 2025 — Class I devices
- Direct marking of the reusable devices
- 26 May 2023 — Implantable and class III devices
- 26 May 2025 — Class IIa and class IIb devices
- 26 May 2027 — Class I devices
- Placing UDI-carriers on the labels of IVDs
- 26 May 2023 — Class D IVDs
- 26 May 2025 — Class C and B IVDs
- 26 May 2027 — Class A IVDs
- Extended transition for legacy devices
- 31 Dec 2027 — class III and class IIb implantable devices (except certain devices for which the MDR provides exemptions)
- 31 Dec 2028 — Other class IIb devices and class IIa, class Im and class Is devices.
- UDI Data: 109 attributes to EUDAMED; New BUDI-DI group concept
- Data Sub: Website entry/upload or electronic XML (via Data Exchange) to EUDAMED
- UDI Label: HRI & AIDC by class (III-2021, IIa/b-2023, I-2025); Direct Mark by class (Label + 2yr)
- STD: GS1/HIBCC/ICCBBA/IFA; SRN; EMDN
- Info: EC Reg, UDI (European Commission)
-
- Ordinance No 128 of 2022-Guideline for Labeling of Codes on Containers to Identify Medical Devices, In Vitro Diagnostics, etc.
- 04 Dec 2019 – PMD Act required barcode labels and registration
- Dec 2022 – Medical devices (except contact lenses), in vitro diagnostics, and consumable materials other than medical devices require barcode labelling based on international standards for immediate containers/wrappings/retail packaging of medical devices.
- Approach: Report device info to database and apply barcode to labels for medical devices and IVDs; future UDI reporting to UDI database
- UDI Data: Device reistration includes: Product Code (DI), Expiry Data, Lot #, Serial #
- UDI Label: HRI and AIDC (1D GTIN-128 barcode symbol or 2D); no Direct Marking now
- STD: GS1
- Info: PMDA (Pharmaceuticals and Medical Devices Agency)
-
- 2019 Apr 08 – UDI Draft Requirements released
- 2019/2020 – UDI Pilot
- 2020 Sep 06 – UDI Formal Requirements (v.3) released
- 2023 Sep 01 – Class B, C, D devices (was 2022 Sept)
- 2024 Sep 01 – Class A
- Database: SAUDI — DI (Ramz)
- Sub: Website upload or Bulk Upload (TBD)
- STD: GS1/HIBCC/ICCBBA
-
- 1 Nov 2022 High-risk implantable (3) All Coronary stents, orthopedic joint replacement implants, and intraocular lens
- 1 Nov 2024 Class D (High-risk) General medical devices and IVDs
- 1 Nov 2026 Class C (Medium-risk) General medical devices and IVDs
- 1 Nov 2028 Class B (remaining) General medical devices and IVDs
- Voluntary Class A (low-risk)
- Database: Singapore Medical Device Register (SMDR) for class B or higher medical devices and the Class A medical device database for Class A medical device. For all risk classes through MEDICS
- Data Sub: Website XLS upload
- UDI Label: HRI & AIDC; Direct Mark
- STD: GS1, HIBBC, ICCBBA
-
- 2018/2020 – Guidelines for Barcodes & UDI Codes
- 2019 Jul 01 – Class IV Devices UDI Labeling req’d
- 2019 Oct – Class IV Devices UDI Labeling req’d
- 2020 Jul 01 – Class III Devices
- 2021 Jul 01 – Class II Devices
- 2022 Jul 01 – Class I Devices
- +1 year – Monthly Supply Report Req’d
- Approach: UDI data reporting and UDI Label by class AND submit Supply Report (Track & Trace) of ~10 distribution metrics each month
- UDI Data: 40 data attributes to IMDIS (Integrated Medical Device Information System)
- Data Sub: website XLS upload /complete data / upload file or machine-to-machine XML transfer via API by Local Rep
- UDI Label: HRI & AIDC, Direct Mark
- STD: GS1/HIBCC/ICCBBA
- Info: MFDS, IMDIS UDI System
-
- Events:
- 2015 -Oct: Administrative Guidance
- 2020 – Nov: UDI Announcement
- Compliance Timing
- Class III implantables: 1 June 2021
- Class III non-implantables: 1 June 2022
- Class II devices: 1 June 2023
- Description
- Approach:new device UDI data labels; similar to FDA model
- Database:Taiwan UDID (TUDID)
- Data: 24 Attributes; similar to FDA (No BUDI)
- Sub: Website upload (Web input) or batch upload
- Label: HRI & AIDC (1D or 2D), Direct Mark
- STD: GS1, HIBBC, ICCBBA
- Info: FDA Medical Devices
- Events:
-
- 2021 Jan 01 – Brexit effective, Guidance released
- 2021 Jan 01 – UK-mfg Class I, Custom, IVD register
- 2021 May 01 – III, II Implantable, Active Implantable, IVD A register
- 2021 Sep 01 – IIB other, IIa, IVD B, IVD Self-test register
- 2022 Jan 01 – OUK-mfg Class I, Custom, IVD register
- 2023 Jul 01 – CE replaced by UKCA mark; Legislation expected to require UDI
- Approach: UK regulatory framework revamped due to Brexit; Mfrs and Devices must be registered in MHRA; GB will NOT comply with EU MDR/IVDR (Northern Ireland will); UDI voluntary field – future UDI legislation will require UDI
- UDI Data: ~70 attributes similar to EU reported to DORS (Device Online Reg. System) includes BUDI-DI
- Data Sub: Website manual entry or XLS upload (1,000 max records); perhaps future machine-to-machine
- STD: GS1/HIBCC/ICCBBA/IFA; GMDN, UKCA
- Info: MHRA Medical Devices (Medicines and Healthcare Products Regulatory Agency)
-
- 2014 Sep – Class III Devices
- Required to bear UDI on the Device
- Contain Standard date format
- Provide UDI on stand-alone software device
- UDI must be submitted to the GUDID database
- 2016 Sep – Class III Devices
- UDI as a permanent marking on the device that are intended to be used more than once and reprocessed before each use
- The FDA does not intend to enforce UDI direct mark requirements for Non-Sterile Class III devices that are manufactured and labeled prior to September 24, 2016, when the device’s UDI can be derived from other information directly marked on the device
- 2016 Sep – Class II Devices
- Required to bear UDI on the Device
- Contain Standard date format
- Provide UDI on stand-alone software device
- UDI must be submitted to the GUDID database
- 2018 – Sep – Class II Devices
- UDI as a permanent marking on the device that are intended to be used more than once and reprocessed before each use.
- The FDA does not intend to enforce UDI direct mark requirements for Non-Sterile Class II devices, other than LS/LS devices that are manufactured and labeled prior to September 24, 2018, when the device’s UDI can be derived from other information directly marked on the device
- 2022 Sep – Class I and unclassified devices, other than I/LS/LS devices
- Required to bear a UDI
- Contain Standard date format
- Provide UDI on stand-alone software device
- UDI as a permanent marking on the device that are intended to be used more than once and reprocessed before each use.
- Directly marked with a UDI
- The FDA does not intend to enforce UDI direct mark requirements for Non-Sterile Class I devices and unclassified devices, including Class I and unclassified device constituents of a co-packaged combination product or kit other than LS/LS devices that are manufactured and labeled prior to September 24, 2022, when the device’s UDI can be derived from other information directly marked on the device
- 2022 Dec – Class I and unclassified devices, other than I/LS/LS devices
- UDI must be submitted to the GUDID database.
- Class I CGMP-exempt devices are excepted from UDI requirements.
- Approach: Report UDI Data & apply UDI to Labels by class
- UDI Data: 57 attributes to GUDID
- Data Sub: Website entry or electronic HL7 SPL (via ESG)
- UDI Label: HRI & AIDC; Direct Mark
- STD: GS1/HIBCC/ICCBBA; DUNS; GMDN/FDA
- 2014 Sep – Class III Devices
We’re Here To Help
Get in touch with us
Let's talk about how DDi can help you