Regulatory
Information
Management (RIM)
for Medical Devices
A modular Regulatory and Process platform for Medical Device / IVD manufacturers to streamline product registration, streamline content processes, manage regulatory impact assessments, automate publishing activities, manage global submissions, and ensure regulatory compliance efficiently.
0+
Countries Supported
0+
Submissions Managed
0%
Part 11 Compliant
0/0
Real-Time Tracking
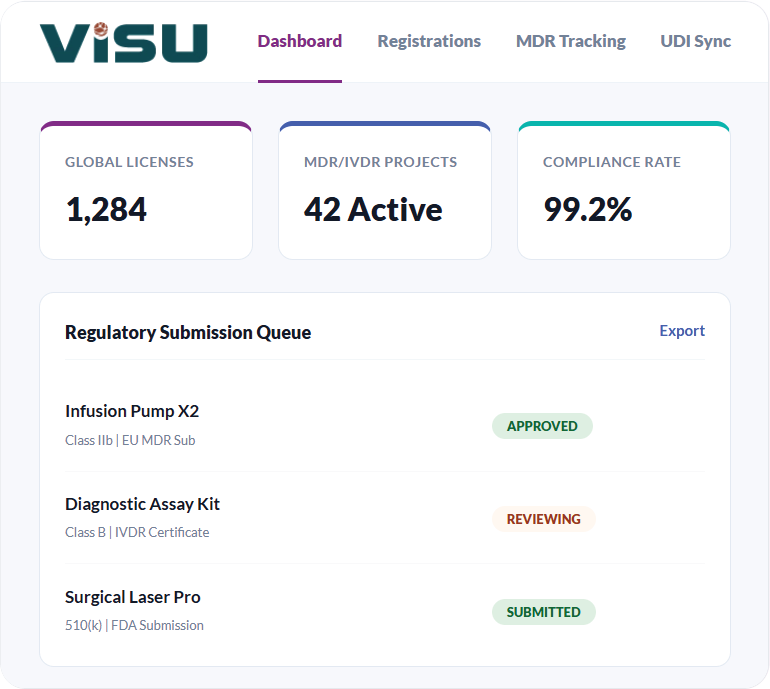
Why Choose Visu RIM?
End-to-end regulatory lifecycle management from planning to tracking to compliance.
Global Visibility
Gain a single source of truth for all product registrations and market authorizations worldwide. Track status
in real-time.
Submission Planning
Efficiently plan and track regulatory activities. Manage submission content plans and timelines to ensure country specific compliance is met.
Compliance Tracking
With built in regulatory requirements for over 140 markets, ensure market compliance at all times and see gaps quickly.
UDI, IMDRF, EU MDR and Others
Built-in support for data standards including UDI, ensuring your data is traceable for future compliance.
Seamless Integration
Leverage built in Document Management module or connect to external EDMS, PLM, ERP, QMS and other systems for a connected ecosystem to exchange Data or Documents.
Advanced Analytics
Dashboard analytics provide insights into submission volume, approval times, and workload distribution across regions.
Key Capabilities
Empowering Regulatory Affairs teams with digital automation.
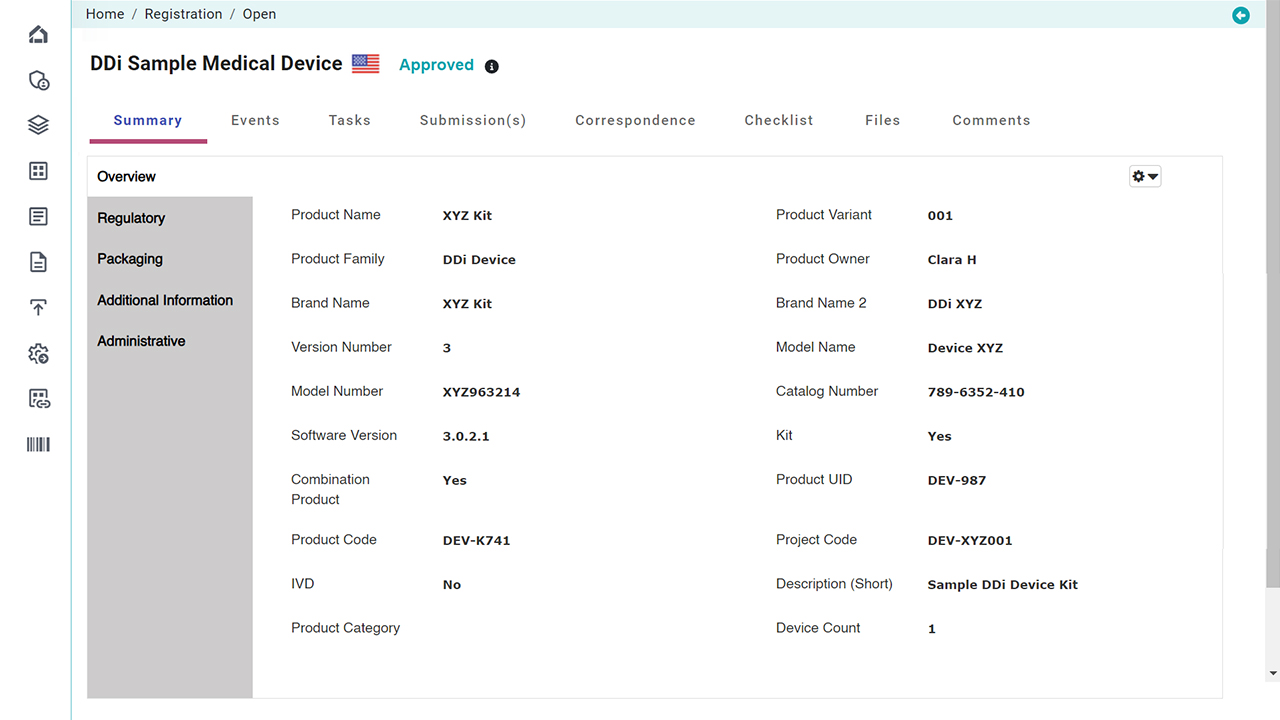
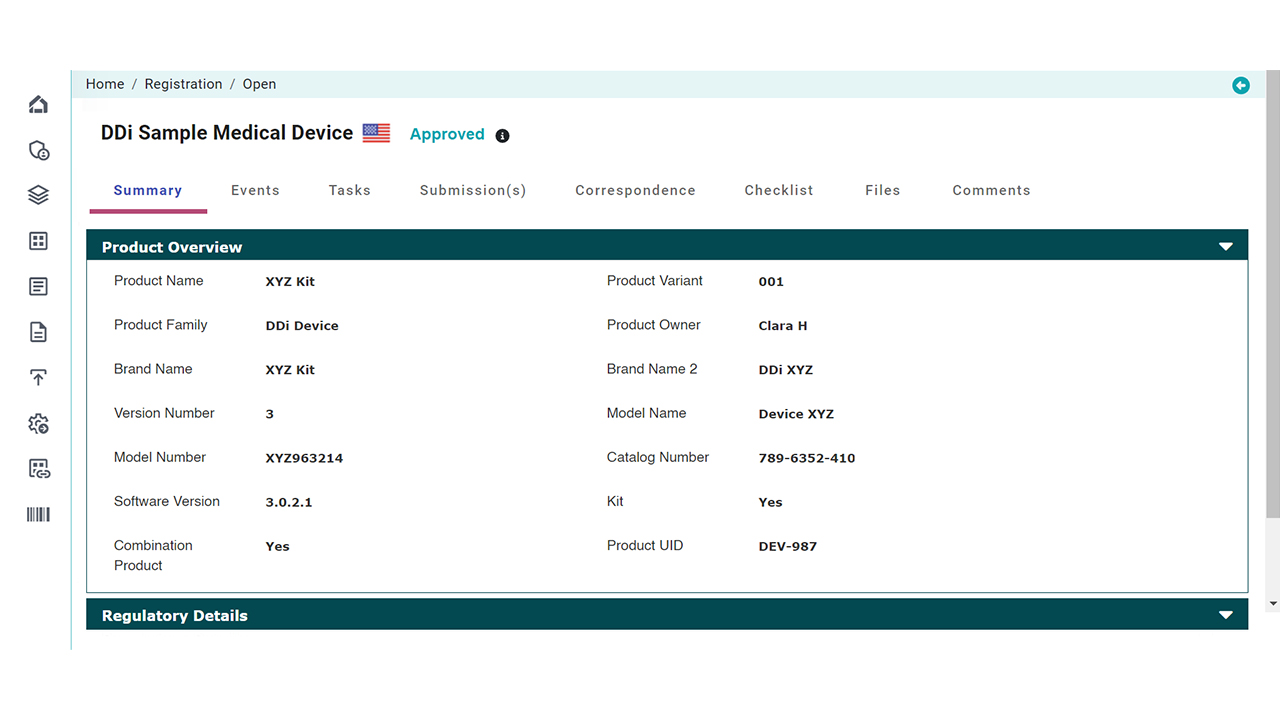
Product Registration Management
Manage the entire lifecycle of product details, certificates, and authorizations across all global markets.
Submission Management
Plan, track, and manage all submission types (Global, FDA, EU MDR, Others) with detailed workflow automation.
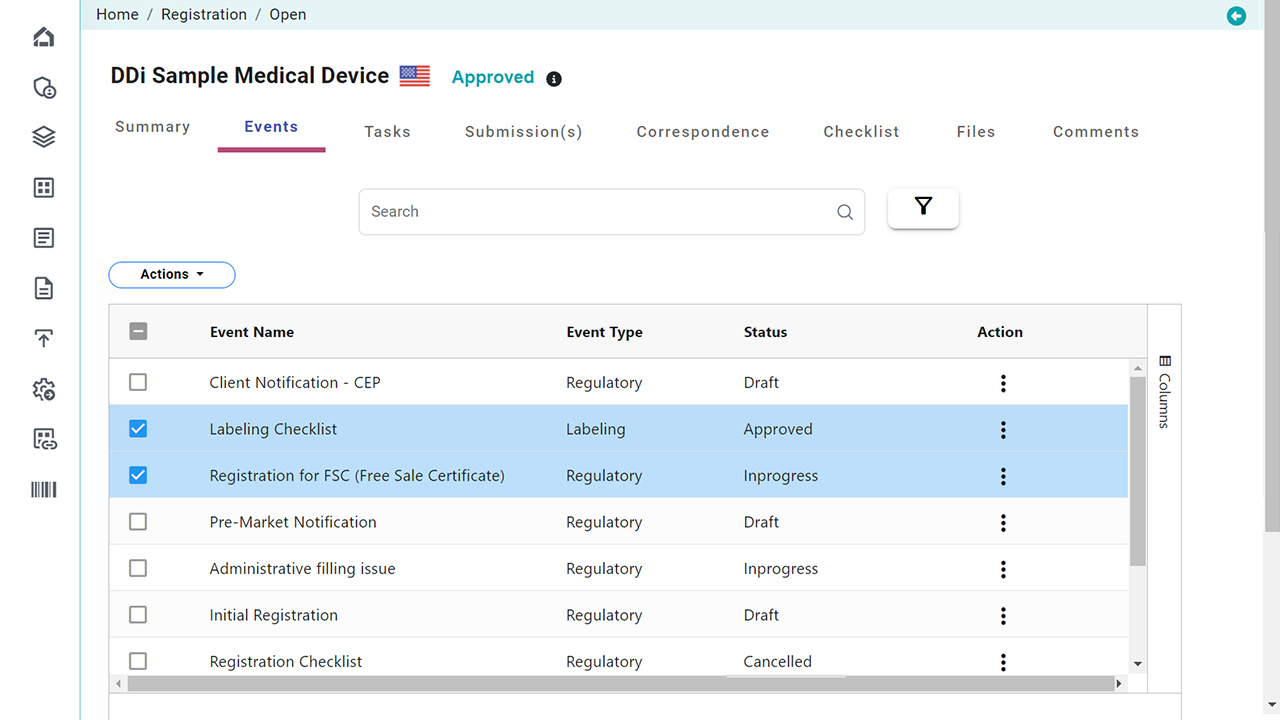
Health Authority Interactions
Centralize all HA correspondence, questions, and commitments to ensure timely responses.
Regulatory Compliance
With 140+ countries regulatory intelligence built in and updated weekly, subscribe to country and function specific alerts.
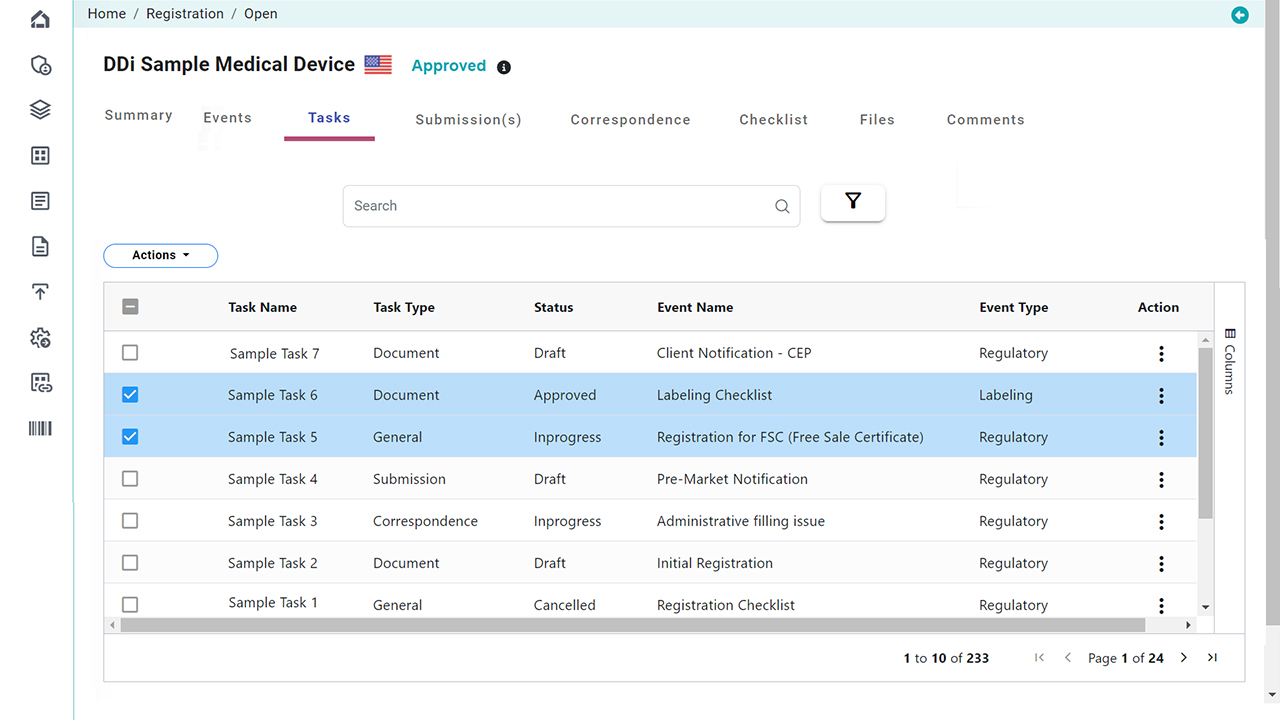
Platform Interface
Check the intuitive interface of Visu RIM, designed for Medical Devices. (Click images to enlarge)








Ready to Transform Your Regulatory Operations?
Move away from manual tracking or spreadsheets. Adopt a compliant, data-driven RIM solution designed for modern MedTech manufacturers.
Tailored Solutions for Your Business
Explore our comprehensive regulatory ecosystem designed for Pharma & Biotech.
Related Tools & Solutions

Regulatory Impact Assessment (RIA)

Submission & Publishing

Product/License Tracking
Explore RIM Insights

Digital RIM for Next-Gen Medical Device Regulatory / Technical...

Regulatory RIM Solution Ensuring Compliance

How Regulatory Systems in Cosmetic industry help ensure Compli...
Let's talk about how DDi can help you