Consumer Health Regulatory Requirements Database for Cosmetics, Supplements & Food
In the evolving landscape of consumer health products, having a reliable and comprehensive Regulatory Requirements Database is crucial for successful product development and market readiness. Whether you’re introducing a new product or managing regulatory changes, access to accurate, up-to-date global requirements enables your teams to plan effectively and remain compliant.
At DDi, our Regulatory Requirements Database for Consumer Health is built to support Cosmetics, OTC, Dietary Supplements, and Food products across multiple regions. We also help teams align with both current and draft guidance documents to better plan short- and long-term regulatory strategies—ensuring clarity, consistency, and compliance from development to post-market.
Our Solution Covers 4 Key Functional Areas:
1. Global Regulatory Requirements Database
Our powerful database includes detailed requirements across functional and business areas such as:
- Clinical trial requirements
- Market launch checklists
- Vigilance and safety/PV protocols
- Testing standards
- Regulatory submission fees and checklists
- Labeling and packaging rules
- Import/export processes
- Renewal and variation timelines
- Full product life cycle requirements
This Regulatory Requirements Database for Food and Consumer Health ensures all teams have centralized access to reliable data and stay aligned across global markets.
2.Regulation & Guidance Document Repository
Gain quick access to global and regional regulatory guidance and draft regulations. Our search-optimized repository simplifies navigation through thousands of documents—saving you time and helping shape strategic decisions based on the latest regulatory intelligence.
3.Ingredients Database
Identify, validate, and manage cosmetic and supplement ingredients across global regulatory frameworks. Our ingredients database supports risk assessment, labeling accuracy, and compliance validation—all tailored to region-specific standards.
4.Regulatory Document Creation & Collaboration
Create regulatory documents using your preferred templates while enabling seamless collaboration. Features include:
- Internal and external stakeholder review workflows
- Access for users without login credentials
- Full document automation (fonts, tables, TOC, headers/footers)
- Output ready for submissions and audits
By centralizing and simplifying access to regulatory intelligence, our platform helps consumer health companies:
- Reduce compliance risks
- Streamline global product launches
- Improve cross-functional efficiency
- Maintain regulatory readiness
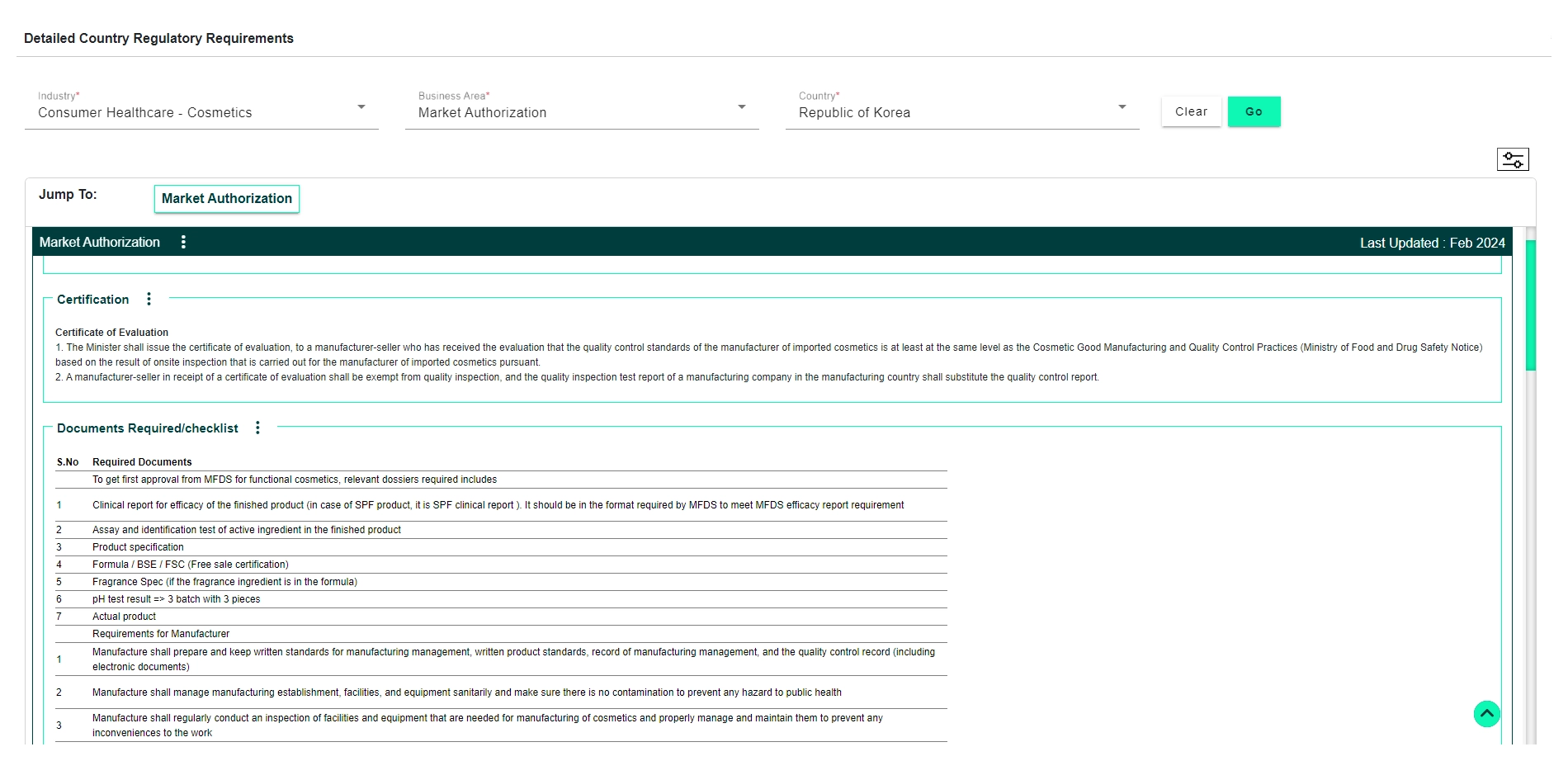
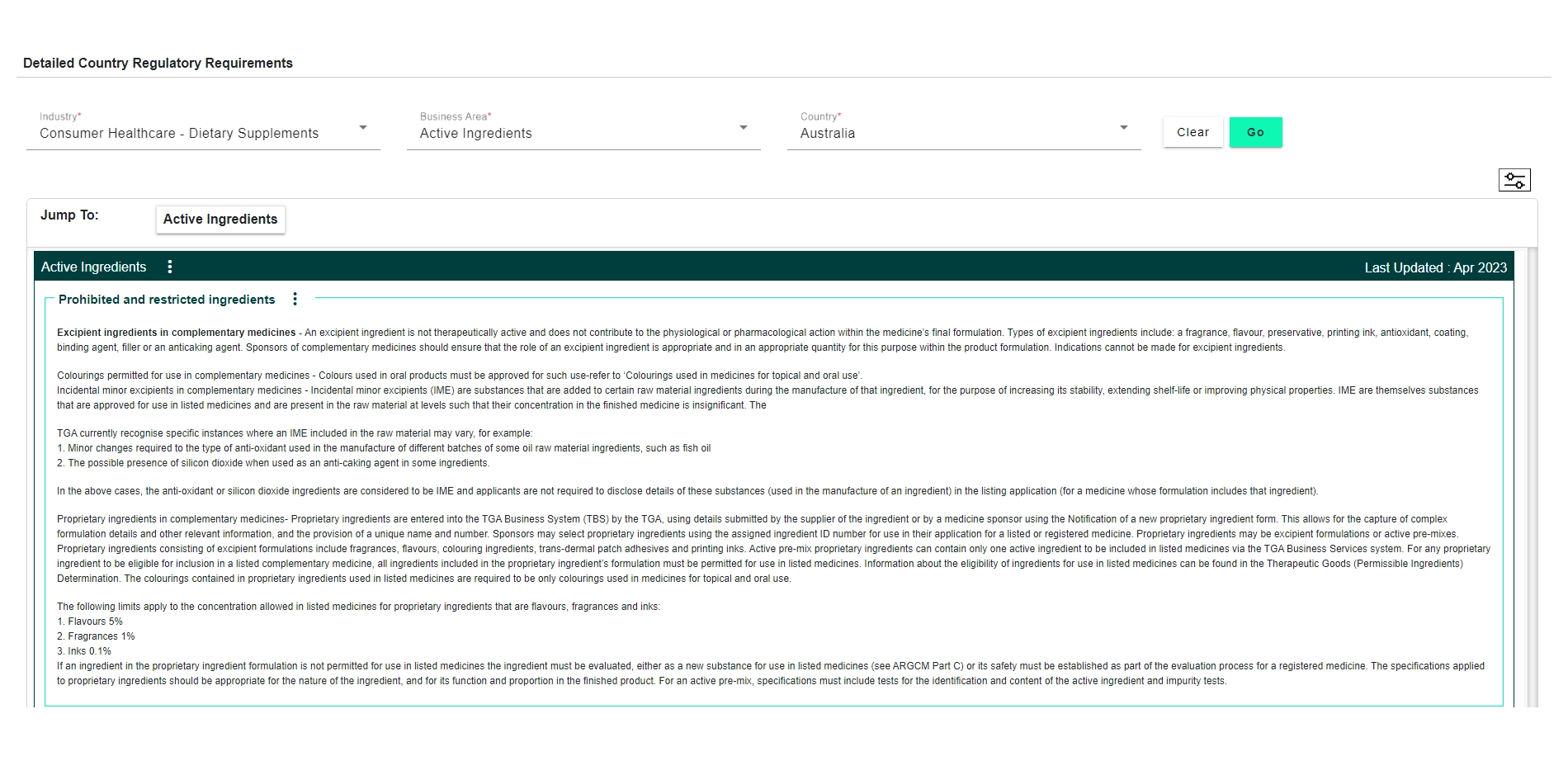
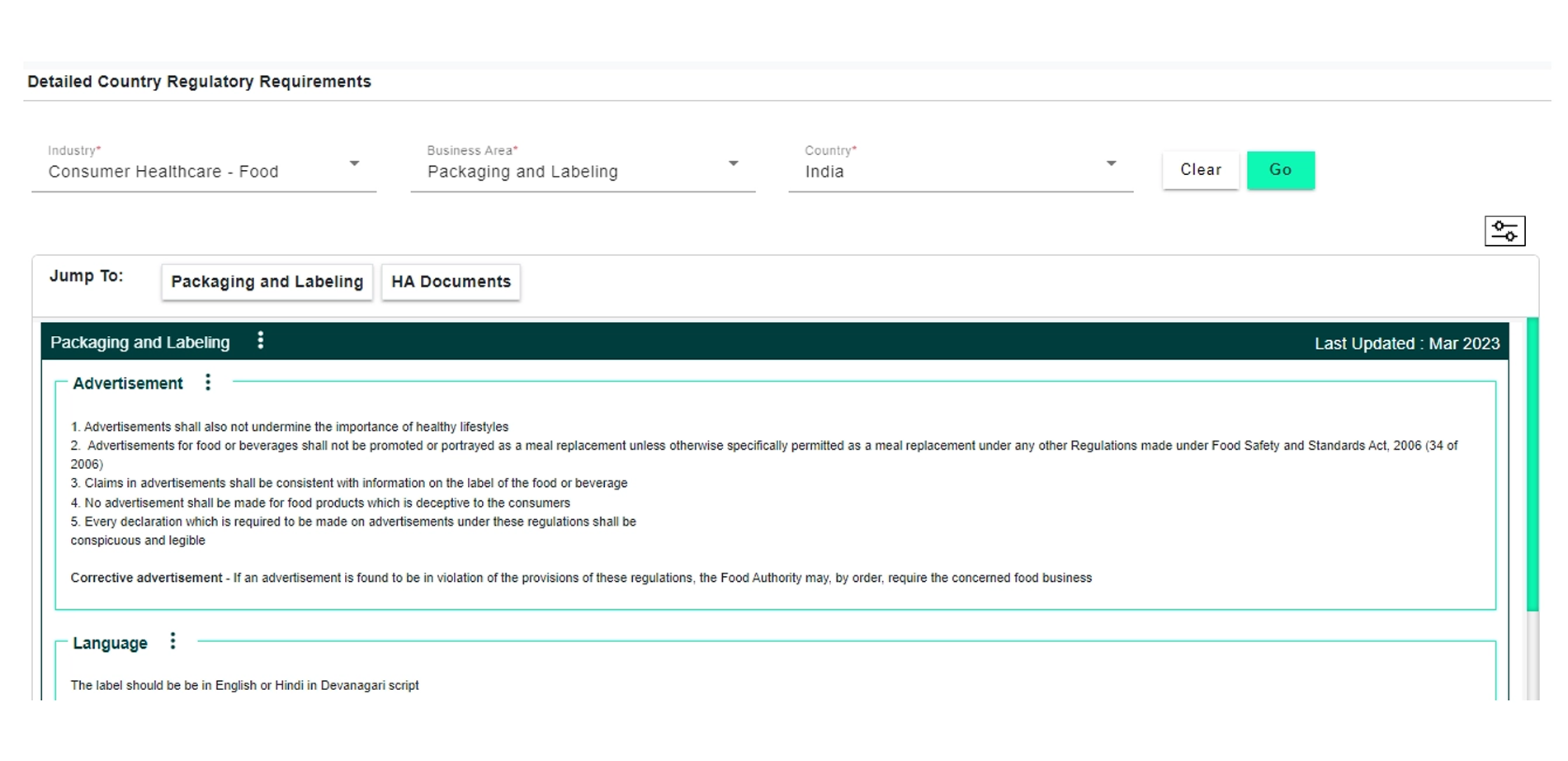
Explore how DDi’s Regulatory Requirements Database for Consumer Health and Food can support your compliance and submission goals.
We’re Here To Help
Get in touch with us
Let's talk about how DDi can help you

