QC Automation of
Labeling / Technical Documents
In the medical device industry, a label is more than just a sticker – it is a critical component of the product. An incorrect expiry date, a missing UDI (Unique Device Identifier), or a mismatched symbol can lead to costly recalls, regulatory fines, and, most importantly, patient safety risks.
Our Automated Label QC Solution leverages AI and statistical methods to eliminate manual, error-prone inspections into a high-precision, automated workflow.
The Challenge: Precision at Scale
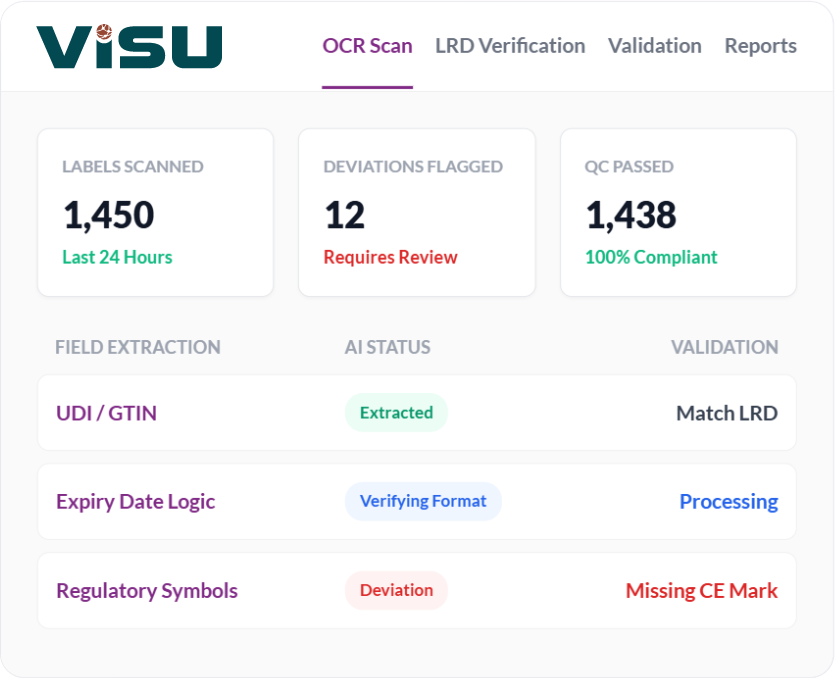
Medical device labeling must adhere to stringent global standards (such as FDA 21 CFR Part 801 and EU MDR).
Manually verifying every field against regulations and product technical documents is slow and susceptible to human fatigue.
Our solution automates this by extracting, verifying, and validating every data point in seconds.
Core Capabilities
Intelligent Field Extraction
Using advanced Optical Character Recognition (OCR) and Natural Language Processing (NLP), the system
scans labels to identify and extract critical data fields, including:
UDI / GTIN
Ensuring the barcode matches the human-readable text.
Lot & Batch Numbers
Tracking production history accurately.
Expiry & Manufacturing Dates
Validating date formats and logic.
Regulatory Symbols
Recognizing CE marks, sterile indicators, and "do not reuse" icons.
Multilingual Instructions
Verifying translations against approved master text.
Validation Against Requirement Documents
The system doesn't just read the label; it understands the "Source of Truth." It automatically cross-
references extracted data against your Labeling Requirement Documents (LRDs) and Master Data files.
Automated Comparison
If the LRD specifies a specific font size or a mandatory warning statement, the AI flags any deviation.
Version Control
Ensure the label being printed is the latest approved version, preventing the use of obsolete templates.
Key Benefits
Reduce Recalls
Eliminate the #1 cause of medical device recalls - labeling errors.
Accelerate Time-to-Market
Speed up the QC bottleneck in the packaging line.
Audit Readiness
Maintain a digital trail of every label checked, complete with time-stamped validation reports.
Ready to Automate Your Compliance?
Don't let a typo trigger a recall. Contact us today for a demo of our AI-driven Label QC platform.
Related Tools & Solutions

Label Content Management

UDI Solutions

eIFU Software for Medical Devices
Knowledge Hub

Is Your eIFU URL Ready for the UDI Database?

Navigating the Global eIFU Patchwork: EU, USA, and the ASEAN P...

Why Unique Device Identification (UDI) Is No Longer “Nice to H...
Let's talk about how DDi can help you