Medical Writing
QC Automation
for Pharma Biotech
With the increase in work load, regulatory expectations and submission timelines getting shorter, quality control for documents has become more complex and resource-intensive. Traditional manual QC processes are no longer sufficient to handle growing document volumes, evolving global regulations, and increasing scrutiny from health authorities.
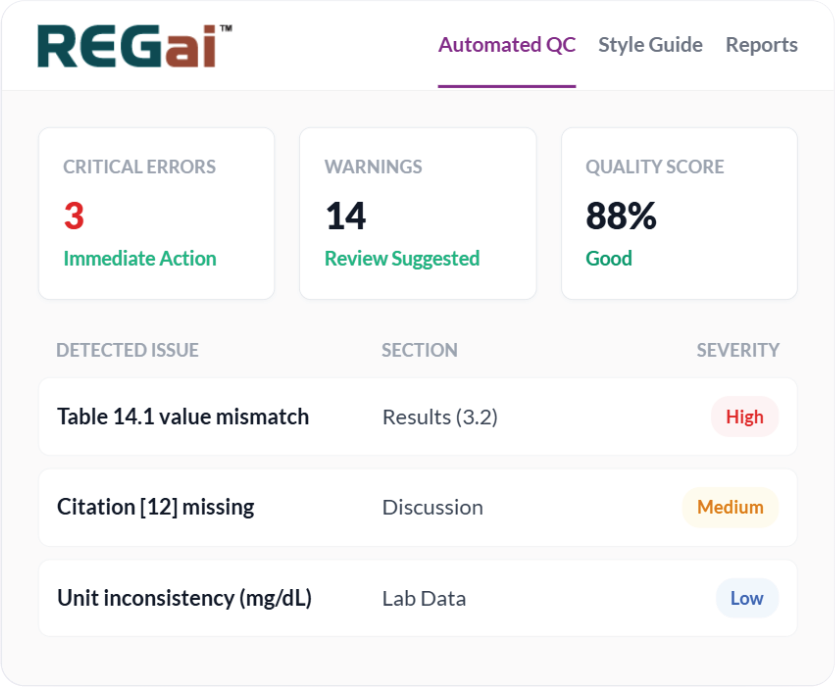
0%
Reduction in QC Time
0%
Consistency Check
0
Formatting Errors
21
CFR Part 11 Compliant
The Challenge: Limitations of Manual QC
At DDi, we understand that QC issues will lead to manual reworks, quality findings, may be submission delays, and additional costs. Even minor misses by QC can trigger issues.
Time-Consuming
Reviewing lengthy documents line by line ( or sometimes side-by-side) slows down submission timelines and increases pressure on medical writers.
Human Error
Fatigue, repetitive checks, and tight deadlines can lead to missed inconsistencies or overlooked formatting issues.
Lack of Standardization
Different reviewers may apply QC rules inconsistently, leading to variable quality across documents and projects.
Scalability Issues
As organizations expand globally, maintaining consistent QC manually becomes increasingly complex.
Our Solution:
Intelligent QC Automation (both AI and non-AI options)
Automation introduces rule-based checks and AI capabilities that systematically validate documents against predefined standards or your rule books
By automating repetitive checks, our platform allows authors and QC reviewers to focus on higher-value work rather than mechanical tasks.
Unlike static systems, our AI models continuously improve by learning
from historical documents, agency feedback, and organizational preferences. This enables the detection of logical gaps, terminology inconsistencies, and conflicting data, ensuring strict alignment with regulatory best practices.
Key Capabilities
Comprehensive validation and rapid scanning for modern Medical Writing.
Template Compliance
Ensure documents follow organizational standards and regulatory expectations every time.
Rapid Scanning
Instantly scan documents for hundreds of parameters that would take hours to review manually.
Comprehensive Validation
Automate checks for data consistency, formatting, style guide adherence, and reference validation.
Table & Figure Verification
Verify in-text data against Tables, Listings, and Figures (TLFs) to ensure 100% data accuracy.
Cross-Document QC
Compare multiple documents (Protocols vs CSRs) to ensure alignment of data and terminology.
AI Learning
Our AI models continuously improve by learning from historical documents and organizational preferences.
Benefits of Automating Your Medical Writing QC Process
Transform your medical writing process with technology-enabled QC.
Improved Accuracy
Automated and AI-driven QC reduces the risk of errors that can lead to regulatory questions or submission rejection.
Faster Turnaround
Automated checks run in minutes, accelerating document finalization and enabling teams to meet aggressive submission deadlines.
Global Consistency
Ensure uniform application of QC standards across teams, geographies, and projects.
Audit Readiness
QC activities are traceable, repeatable, and documented, supporting inspection preparedness and quality audits.
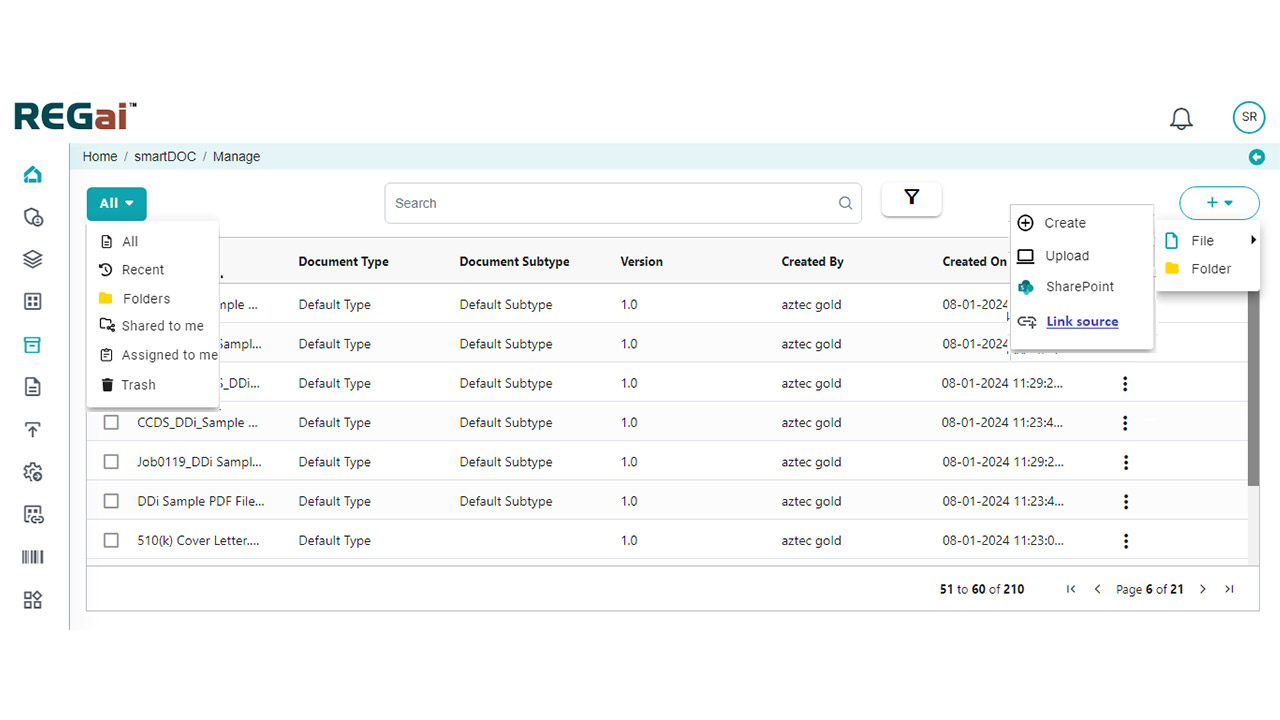
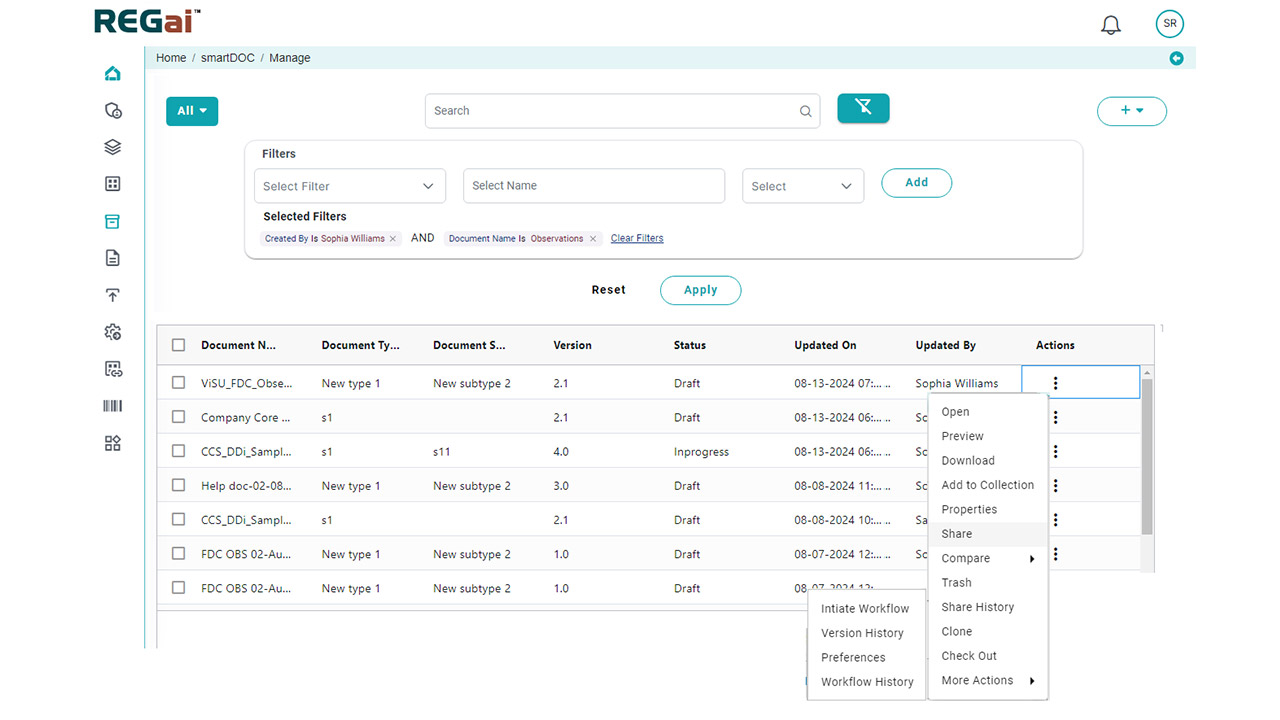
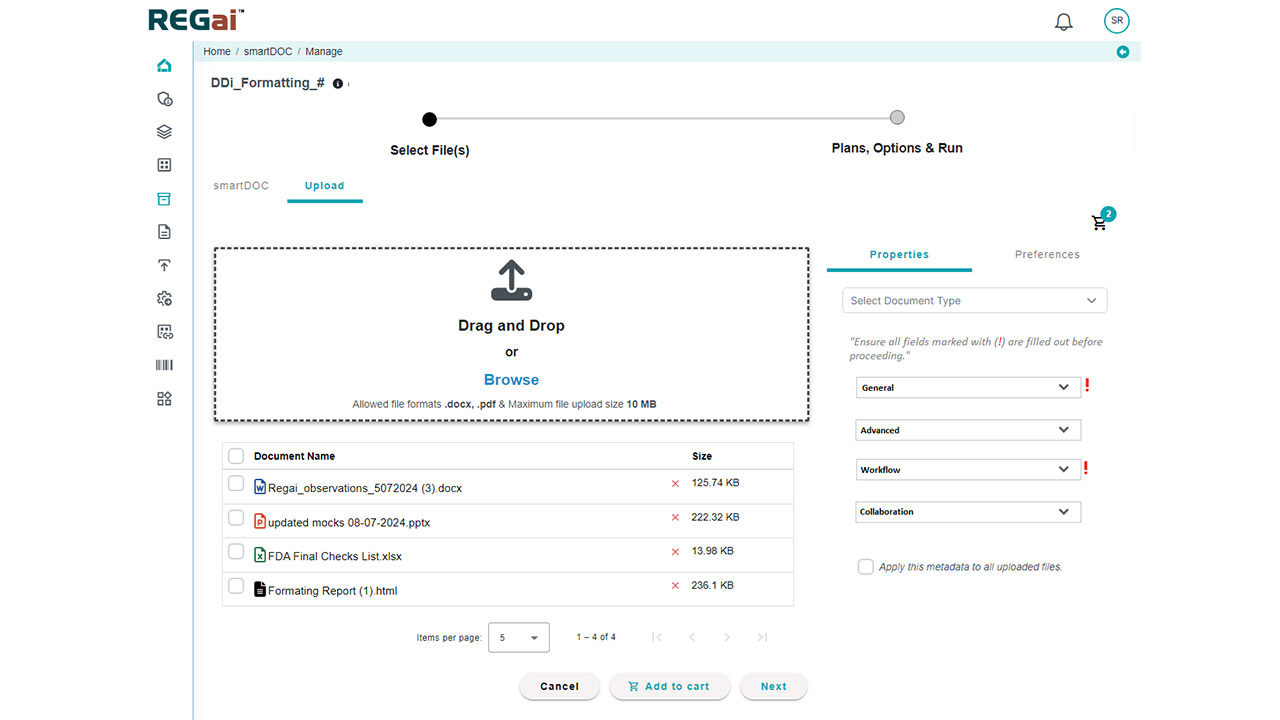
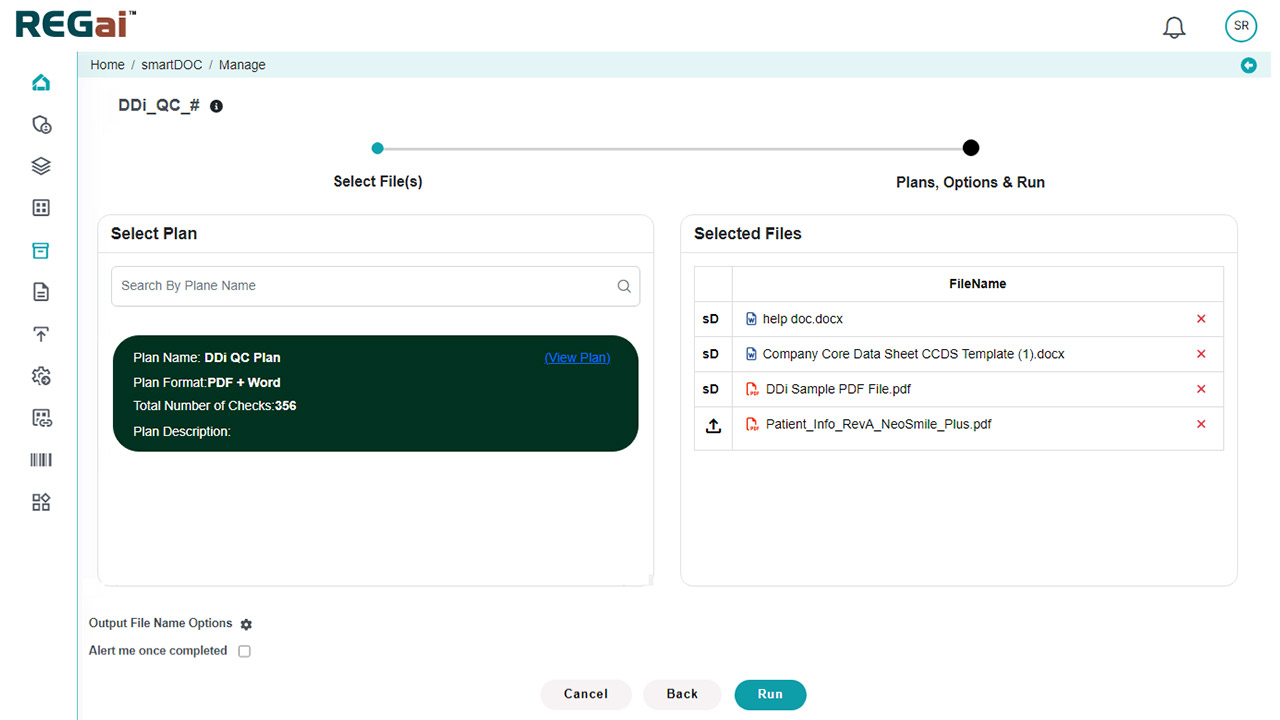
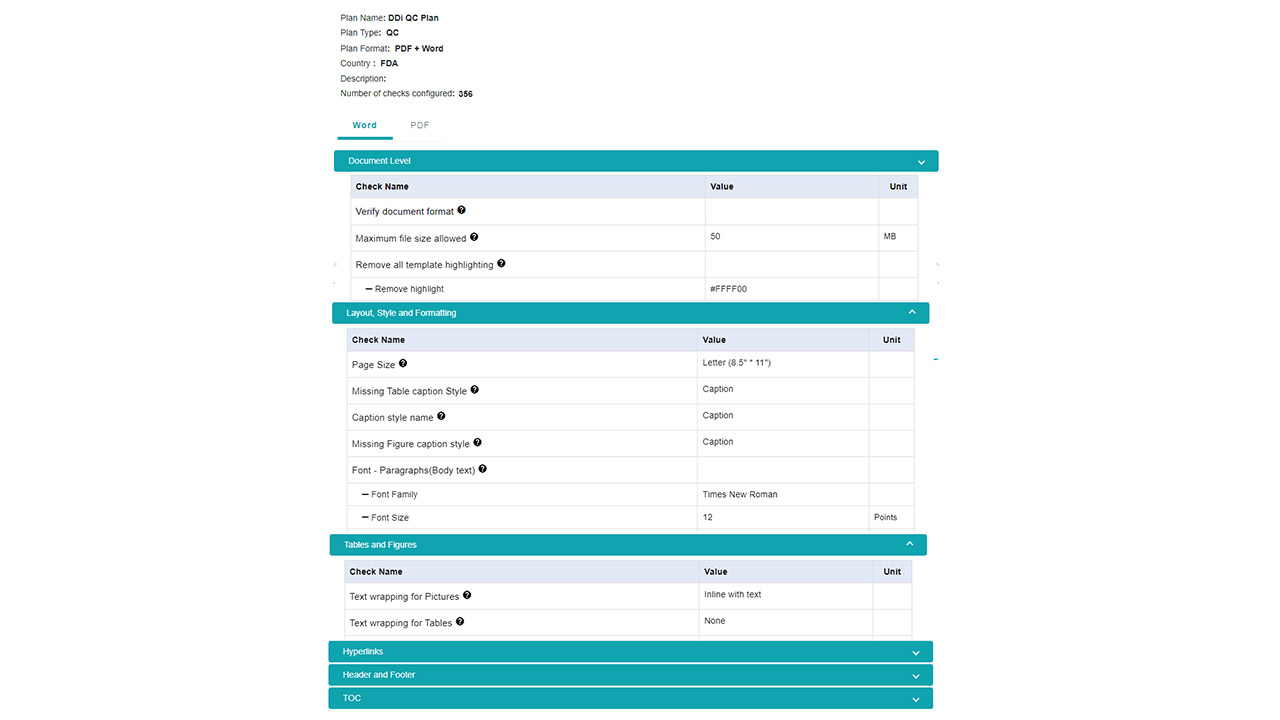
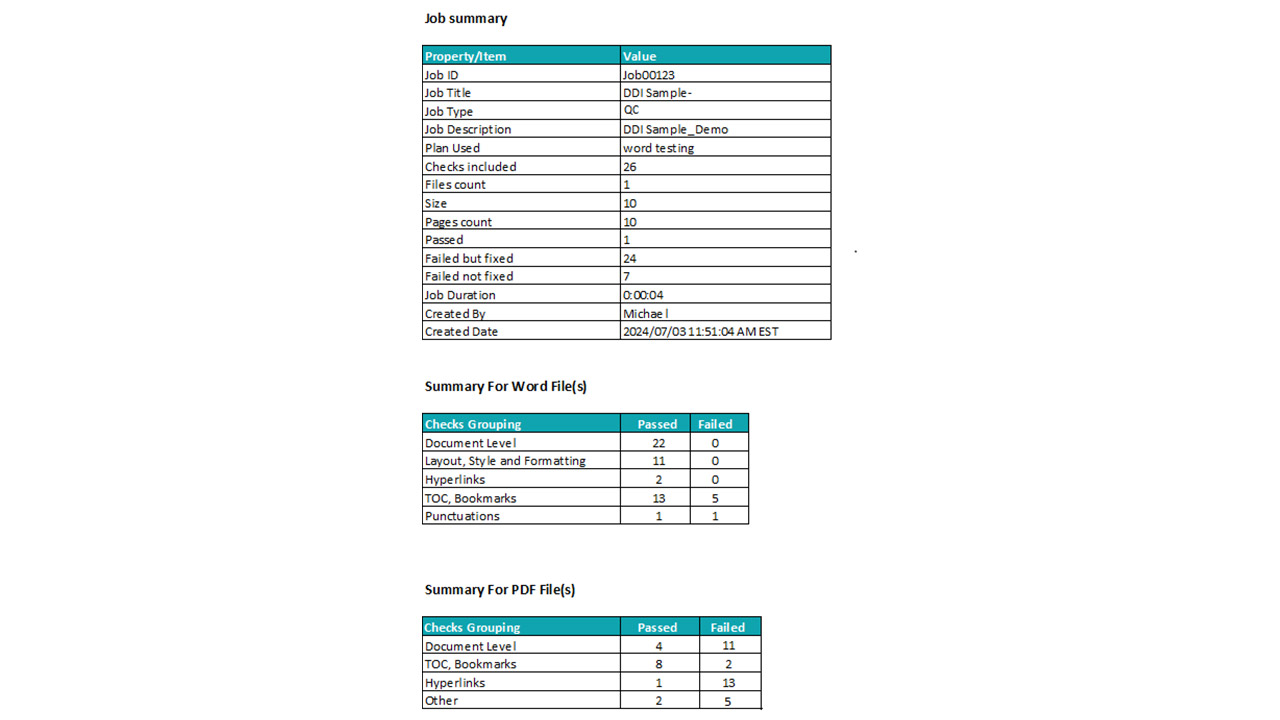
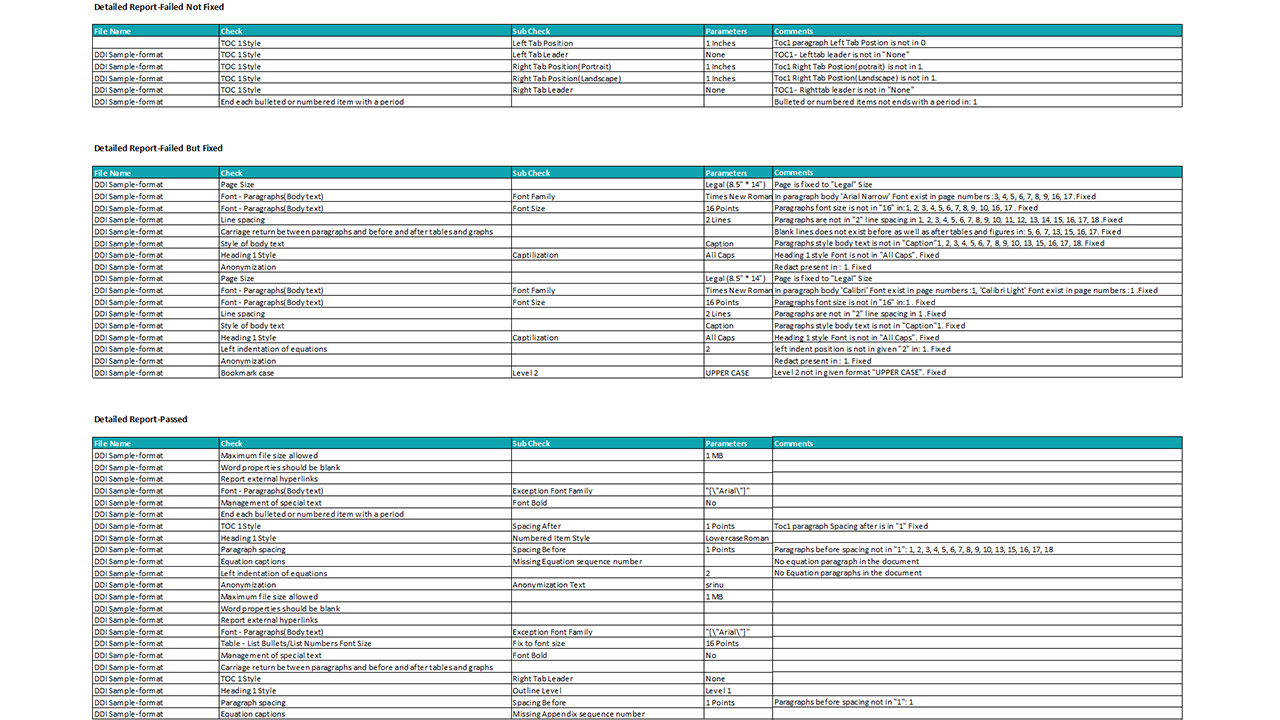
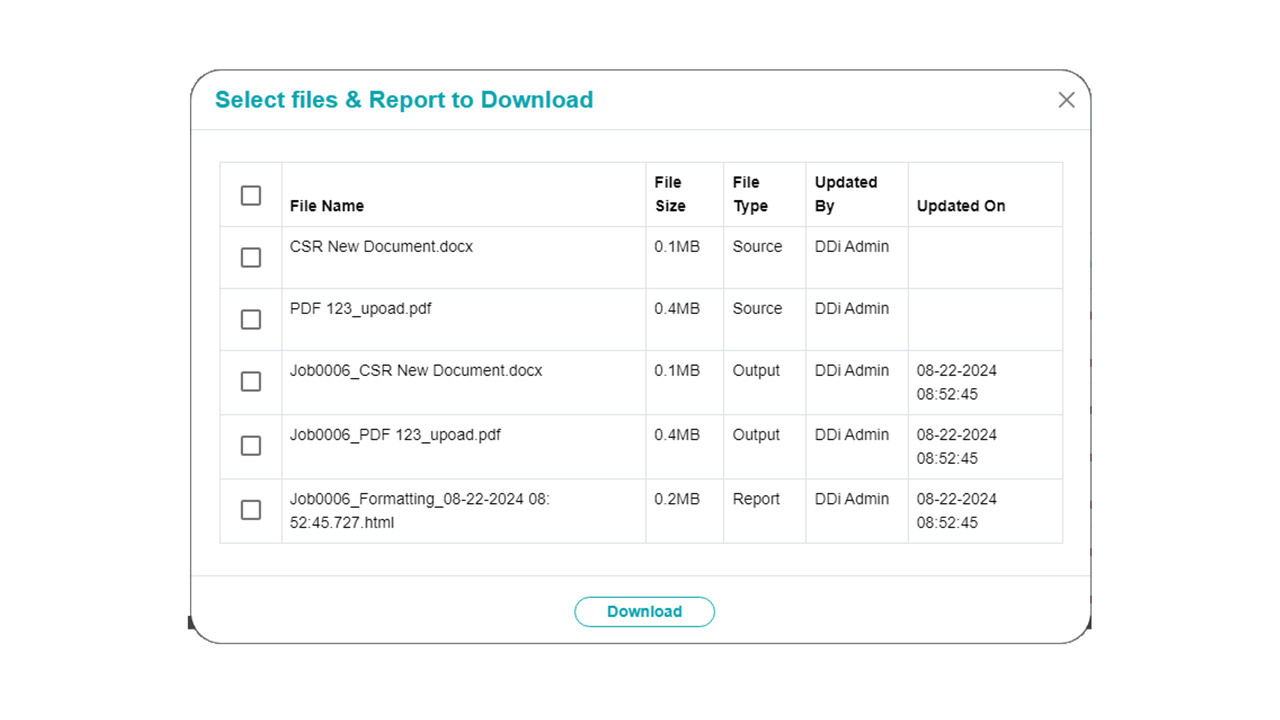
Platform Interface
Experience the intuitive design of DDi's Medical Writing QC Automation process.








Ready to Transform Your QC Process?
Move away from manual redlining. Adopt an AI-driven, automated QC solution designed for modern Medical Writing.
Related Tools & Solutions

Regulatory Impact Assessment (RIA)

Medical Writing Formatting Autom...

Medical Writing Solutions
Explore Insights

Why CMC Authoring Is the Biggest Drug Development Bottleneck: ...

The Future of Medical Writing: How AI Agents Are Redefining Cl...

Expert Medical Writers Versus Automation
Let's talk about how DDi can help you