CMC Authoring
Solutions
In today’s rapidly evolving product development environment, the need for accurate, compliant, and high-quality CMC documentation has never been greater.
DDi’s CMC Authoring Solutions use advanced AI technology to modernize traditionally manual and time-consuming documentation processes into a faster, smarter, and highly efficient workflow.
At the center of our solution are the Authoring Agent and QC Agent. These intelligent agents act as digital collaborators for your CMC teams. The Authoring Agent helps generate high-quality first drafts, context-driven content, and concise summaries. The QC Agent automates comprehensive quality checks across content, data, and document formatting.
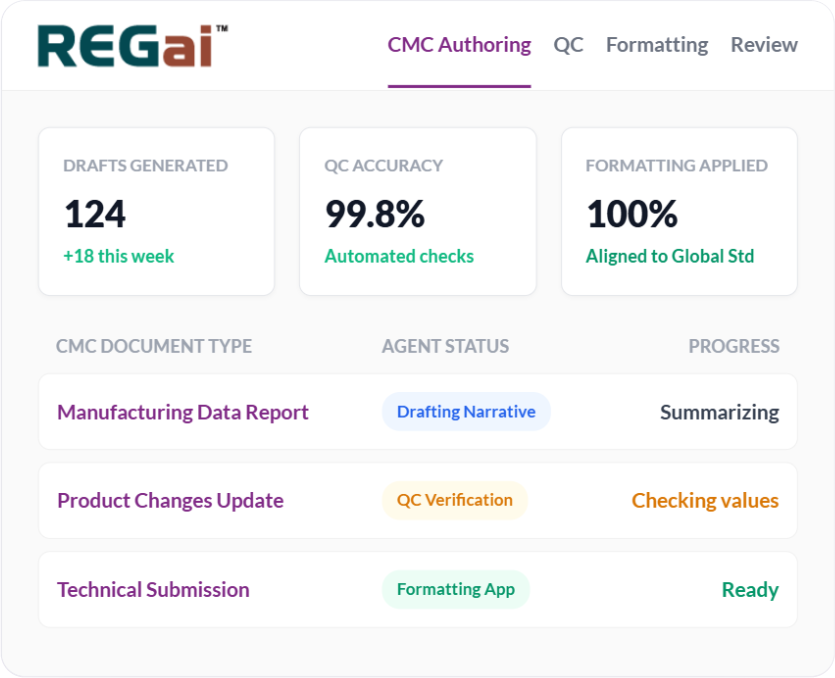
Authoring Automation
Modern CMC documentation requires more than templates. It requires intelligence. Our Authoring Agent
creates draft sections for human review, allowing teams to spend less time starting from scratch and more time
refining technical content and strategic messaging.
By integrating AI-powered writing agents, we simplify the most resource-intensive parts of document development
Automated Summarization
Generate summaries of technical reports, product changes, manufacturing data, and regulatory updates instantly, ensuring critical insights are never missed.
Context-Aware Assistance
Receive real-time AI recommendations to enhance existing content, improve clarity, and maintain consistency across documents.
Data-to-Text Generation
Transform structured manufacturing and quality data into well-written document sections, reducing repetitive manual drafting..
Automating Quality Control (QC)
Even minor errors in CMC documentation can delay approvals and submissions. Traditional QC often depends on
multiple manual review cycles, increasing timelines and effort.
DDi’s QC Agent brings speed, consistency, and objective precision to document review.
Consistency Checks
Ensure terminology, values, abbreviations, and references remain consistent across lengthy dossiers and multiple files.
Compliance Verification
Automatically compare content against global regulatory expectations and internal quality standards.
Faster Review Cycles
Identify issues early and recommend corrections automatically, helping teams achieve right-first-time submissions.
Formatting Automation
Final document formatting often becomes a major bottleneck before submission. DDi’s Formatting Agent
removes the burden of manual styling and layout corrections.
Global Standard Alignment
Automatically format documents to meet submission standards for agencies such as FDA, EMA, and other global authorities.
Template Automation
Apply company templates, branding rules, headers, footers, tables, and styles instantly with precision.
Error-Free Output
Detect and correct layout issues that are commonly missed during manual checks, delivering polished and submission-ready documents faster.
Ready to Transform Your CMC Documentation?
Do not let manual processes slow down your product timelines. Experience the next generation of CMC document automation with DDi’s Agentic AI solutions.
Related Tools & Solutions

Medical Writing Formatting Automation

Medical Writing QC Automation

Medical Writing Solutions
Explore Insights

Why CMC Authoring Is the Biggest Drug Development Bottleneck: How T...

The Future of Medical Writing: How AI Agents Are Redefining Clinica...

Expert Medical Writers Versus Automation
Let's talk about how DDi can help you